Introduction
Heterotopic ossification (HO) is defined as the development of mature bone tissue outside where normal bone should exist [1]. HO is known to occur under certain conditions, including fractures, dislocations, orthopedic surgeries, and post-burn in-jured tissue [2–5]. While HO can present anywhere in the body, the most commonly affected areas are the pelvis, thigh, shoulder, and elbow, which are prone to trauma [6]. Clinical manifestations can vary widely, from presenting no symptoms at all to joint stiffness or pain [3–6]. The early stage of HO is also called the inflammatory phase, where the affected site often exhibits swelling or tenderness on physical examination. In this early phase, HO grows rapidly and can be misdiagnosed as soft tissue sarcoma [7]. As the growth rate decreases in the late phase, the bone formation settles and hardens. This localized bone is firm and often restricts joint motion. Although pressure sores accompanied by HO have been reported, there have been no reports of osteomyelitis (OM) occurring in HO [8]. Herein, we report a rare case of a 60-year-old man who developed OM in HO after reconstruction of a trochanteric pressure sore. Ethical approval for this study was obtained from the Institutional Review Board of Seoul National University Bundang Hospital (IRB No. B-2203-746-103). Considering the retrospective nature of the study, patient informed consent was waived. This research is registered to Clinical Research Information Service.
Case
A 60-year-old male with paraplegia presented with a pressure sore on his right but-tock. He had undergone reconstructive surgery at another hospital 8 years prior; the lesion was a grade 4 pressure sore with underlying muscle exposure and surrounding dead space. Extensive debridement was performed, and the soft tissue defect was reconstructed using a tensor fasciae latae advancement flap. The patient was discharged 4 weeks after surgery without major problems.
Eleven months after surgery, the patient visited the outpatient clinic with a wound opening over the reconstructed flap and persistent purulent discharge. Though he had been ad-ministered oral antibiotics for 4 weeks at a local hospital, there was no improvement. Methicillin-resistant Staphylococcus au-reus (MRSA) was identified in the pus culture, and the patient was treated with intravenous vancomycin for 2 weeks in another hospital. However, the level of C-reactive protein had increased with no improvement in clinical infection signs; therefore, the patient was admitted for wound evaluation. Computed tomography (CT) showed HO lesions over the muscular layer, and the surrounding soft tissue was diffusely infiltrated and more edematous (Fig. 1). Therefore, surgical debridement of infectious tissue was planned. Infected soft tissue and pus formation around the HO were observed intraoperatively. HO lesions were removed, and the surrounding infected soft tissue was completely debrided. Fortunately, the infectious tissue was confined to the soft tissue layer, and we were able to confirm the viability of the surrounding tissue after debridement. Su-prafascial dissection was performed, and the wound was closed using a bilateral advancement flap (Fig. 2). Bone marrow was exposed in some portions of the HO lesions, which were suspected to have marrow infection with sequestration on gross examination (Fig. 3). Histopathological examination confirmed that inflammatory cells had infiltrated the marrow of the HO lesions (Fig. 4). The culture result of the removed HO revealed MRSA. We confirmed the absence of infection in the underlying bone and complete removal of the infected tissue and HO. The elevated level of C-reactive protein normalized on the 5th postoperative day and there was no sign of infection in the wound; therefore, at the end of the first week after the operation, intravenous antibiotics were discontinued. The patient was discharged without complications. At 6-month follow-up, the wound was well healed and there were no signs of recurrence.
Fig. 1.
Computed tomography findings of trochanteric heterotopic ossification. Computed tomography shows new bone formation (yellow arrows) in the soft tissue layer of the reconstructed area, while the surrounding tissue is diffusely infiltrated and edematous. (A) Axial view. (B) Sagittal view.
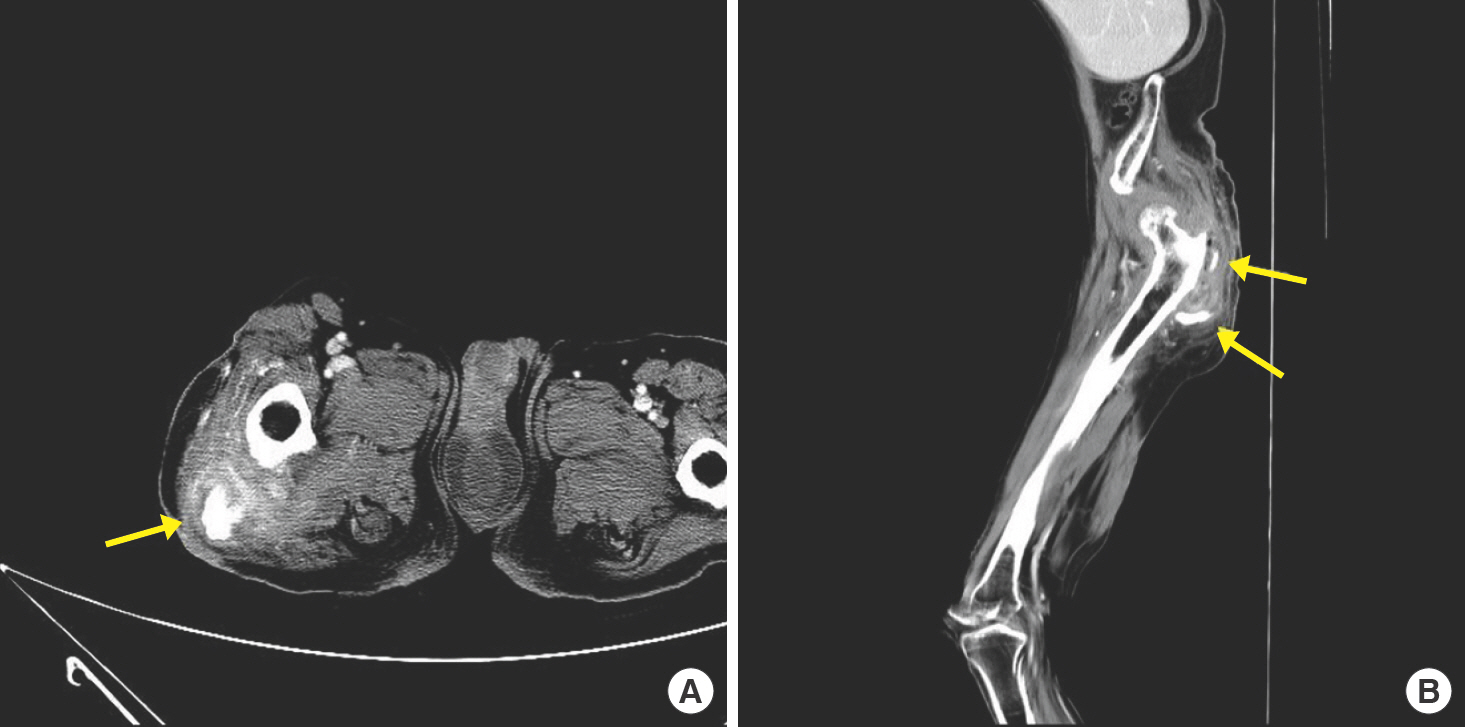
Fig. 2.
Medical photographs of the patient. (A) Initial preoperative photograph before the first operation. Grade 4 pressure sore with underlying muscle exposure and surrounding dead space could be seen. (B) Postoperative photograph taken immediately after the second operation performed using bilateral advancement flap. (C) Postoperative photograph taken 6 months after the second operation. The wound was well healed and there were no signs of recurrence.
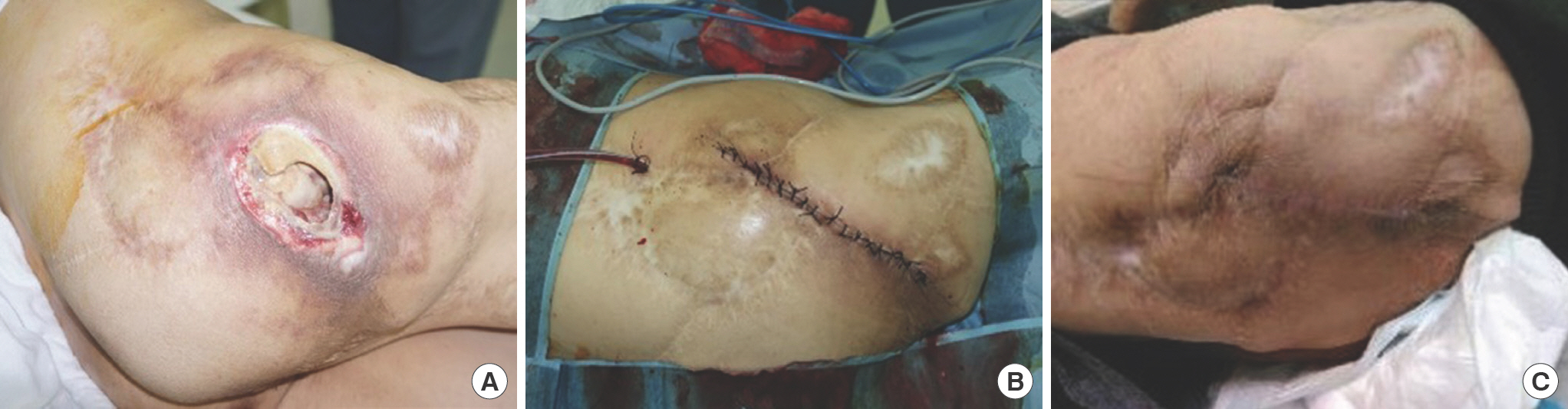
Fig. 3.
Gross findings of intraoperatively resected heterotopic ossification. Bone marrow was exposed in some portion of the heterotopic ossification lesions, which was suspected to have marrow infection (yellow arrow).
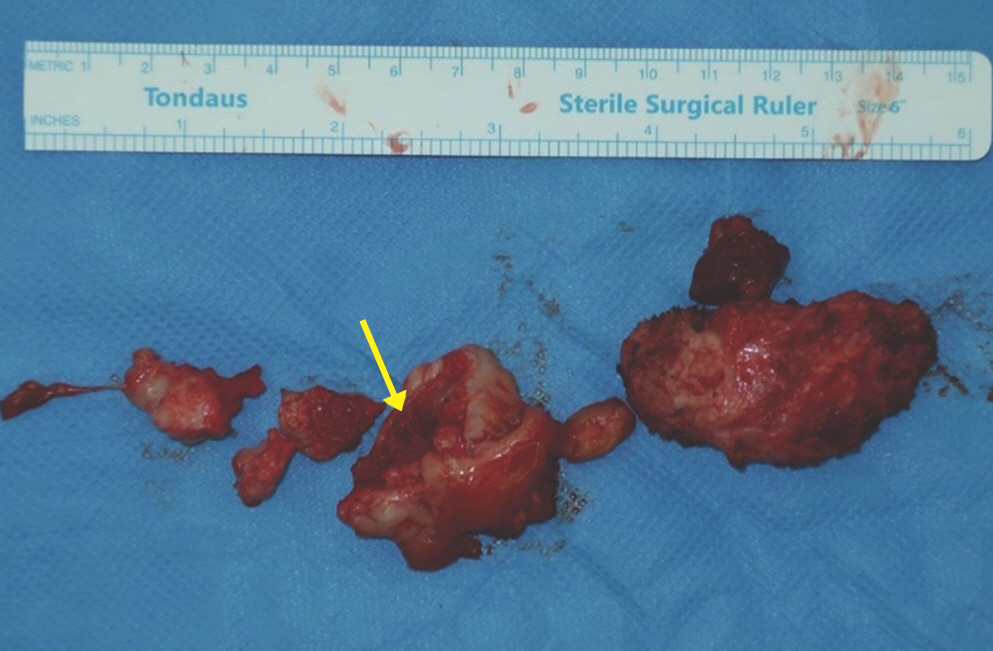
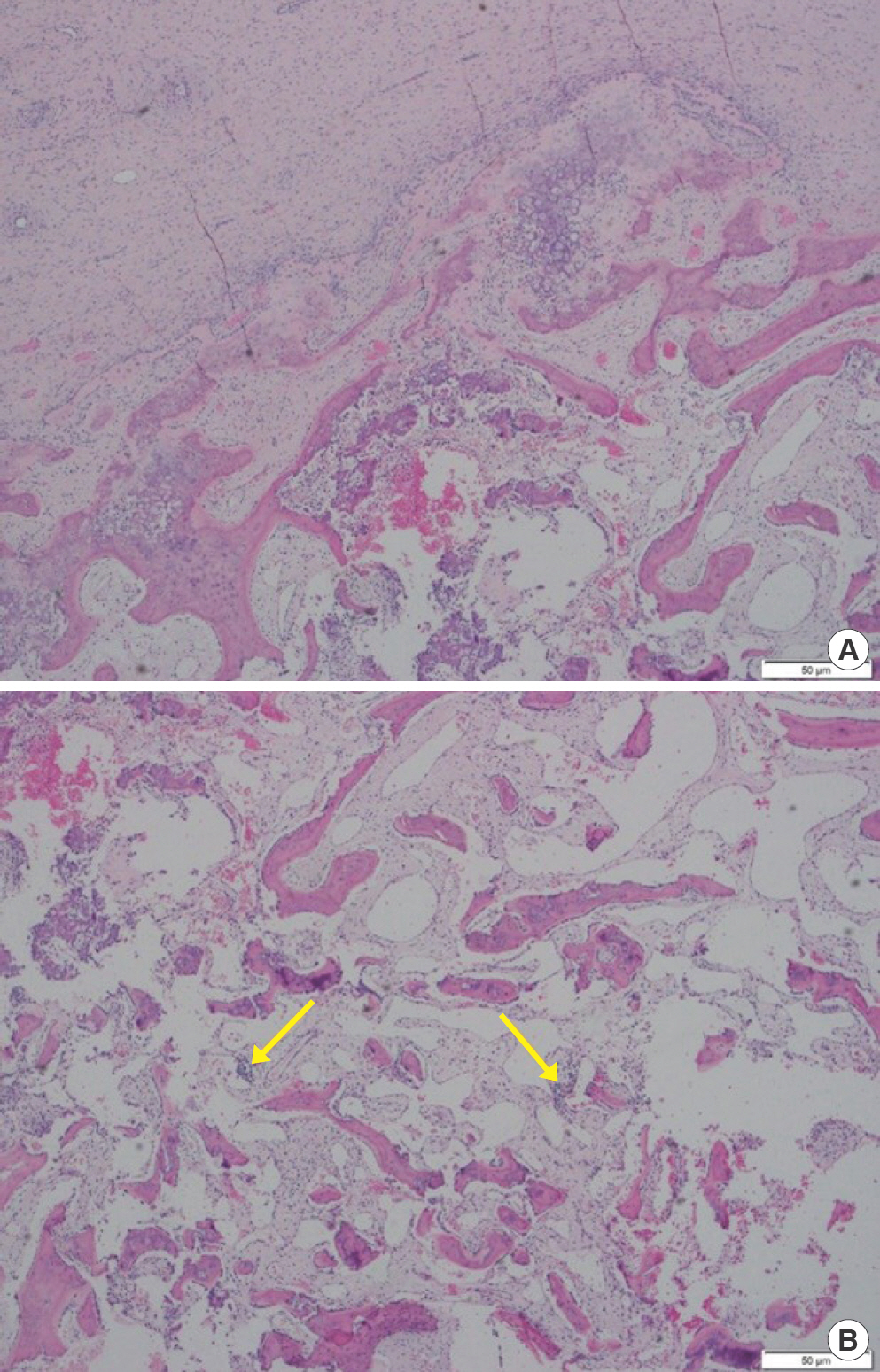
Fig. 4.
Histopathologic findings. (A) Cartilage and cortical bone were lost, leaving only boundaries. Additionally, numerous inflammatory cells were observed in the surrounding fibrous tissue, sug-gesting osteomyelitis (H&E stain, ×40). (B) Inflammatory cells (yellow arrows) are identified without any fat or hemopoietic cells inside the bone marrow, indicating chronic osteomyelitis (H&E stain, ×40).
Discussion
Some predisposing circumstances such as orthopedic surgery or trauma are known to increase the likelihood of HO [6]. When clinically suspected, HO is initially diagnosed by radio-logic examination, and CT findings show characteristic zonal maturation of the HO lesions [9]. In the early phase, HO may present as a low-density mass-like lesion on CT, for which the possibility of cancer must be ruled out. If the likelihood of cancer is clinically low, the diagnosis of HO may become more apparent over time as ossification progresses; during the course of several weeks, radiographic maturation presents as well-defined peripheral ossification [7]. Patients with paraplegia have a higher risk of HO, and pressure ulcers also increase the risk of HO as damaged soft tissue is more susceptible to ectopic bone growth [10,11]. As our patient also had paraplegia, it could be assumed that the inflammatory reaction in damaged tissue after the flap operation created an environ-ment conducive for the development of HO.
OM is an infection-related inflammatory disorder of the bones and is generally categorized as acute or chronic. Such classification is typically based on histopathological findings rather than the duration of the illness [12]. Management of OM is problematic because the method and purpose of treatment vary according to the clinical situation. Antibiotic treatment for at least 6 weeks is usually required, and surgical debridement is often performed to remove infectious lesions [13]. OM can develop in the coccygeal or trochanteric bone adjacent to pressure sores [12,13]. The standard diagnostic tool for pressure sore-related OM is a histological examination of bone biopsy specimens [14]. Histopathologic examination will reveal inflammatory cell infiltration in the bone marrow, while common findings in pressure sore-related OM include reactive bone formation and broad fibrosis.
The current report presents a very rare case of HO accompanied by OM. Though there have been reports of HO in pressure sores or manifestation of OM in pressure sore areas, this is the first report of OM in HO that occurred after surgical reconstruction of a trochanteric sore [10,12,13]. Although HO is not commonly encountered in clinical settings, we emphasize that infection may occur in the newly formed bone. Unlike other OM, in this clinical situation complete removal of the infectious source is possible; therefore, surgical debridement is the most effective treatment. Histological findings, such as infiltration of inflammatory cells in the bone marrow, were con-sistent with chronic OM. Although the order of occurrence of OM in relation to the infected surrounding tissue is unknown, one assumption is that the antibiotic treatment prior to surgical removal showed no response due to OM of HO. This is supported by the fact that the patient recovered immediately after simple debridement and HO removal in the second operation. Long-term antibiotic treatment, which is generally used to treat OM, was ineffective because antibiotics could not effectively reach the marrow of the HO [15]. In such clinical conditions, surgical removal of the HO lesions is the most effective treatment for chronic infection.
In conclusion, although OM from HO is a rare phenomenon, it should be considered in patients presenting with persistent signs of infection that do not respond to antibiotic treatment. If persistent intravenous antibiotics use fails to control infection, it is important to verify on CT or magnetic resonance imaging the presence of accompanied HO. Even HO enclosed over cortical bone or cartilage could be infected and biopsy and culture are recommended to determine if it has proceeded to OM. Complete removal of HO accompanied by OM is possible with less damage to underlying normal structures such as bone, and is very effective for wound and infection control. Diagnosis of OM from HO is important in that it can determine the direction of future treatment.















