 |
 |

|
| Aims and Scope |
| About the Journal |
| About the Society (KWMS) |
| Editorial Board |
| Open Access |
| Principles of Transparency and Best Practice |
| Editorial Office |
| Subscriptions |


|
| Aims and Scope |
| About the Journal |
| About the Society (KWMS) |
| Editorial Board |
| Open Access |
| Principles of Transparency and Best Practice |
| Editorial Office |
| Subscriptions |

AbstractBackground Split-thickness skin graft (STSG) is a fundamental and versatile technique used in reconstructive surgery. Despite numerous studies addressing the management of STSG donor sites, there is still little consensus on the optimal dressing method. This study aimed to analyze the effectiveness of a novel method that combines a fenestrated polyurethane film with negative-pressure wound therapy (NPWT) over conventional moist dressings.
Methods Between January 2021 and December 2022, 23 patients who received STSG were retrospectively reviewed. In group A, a polyurethane film punctured with a 20-gauge needle was applied to the skin graft donor site. A NPWT system (CuraSys) was subsequently applied over the film. The conventional NPWT was replaced with a portable NPWT (PICO system) on the day of discharge. On the 14th postoperative day, the NPWT and film were removed. In group B, patients were administered a traditional moist dressing until epithelization. Postoperative pain, reepithelization rate, and dressing cost per cm² were compared.
Results Group A exhibited a lower mean pain score on all the measured days. Furthermore, group A demonstrated a significantly shorter time required for complete reepithelization (18.07±8.43 days vs. 28.20±5.25 days, P=0.003) and a lower dressing cost per square centimeter (6,517±6,162 KRW [4.82±4.56 USD] vs. 23,036±27,157 KRW [17.05±20.11 USD], P=0.006).
IntroductionSplit-thickness skin graft (STSG) is a fundamental and versatile technique within the domain of reconstructive surgery, widely employed to address a diverse array of skin and soft tissue defects. By harvesting a portion of the epidermis and a variable thickness of the dermis from a donor site, STSG allows the transplantation of healthy skin to the skin defect area caused by trauma, tumor excision, or burns, thereby facilitating wound closure and promoting optimal healing [1].
Donor sites for STSGs often demonstrate significant morbidity [2]. Harvesting the skin from these areas may result in pain, delayed wound healing, infection, and scarring, all of which can affect the patient’s quality of life and prolong the overall recovery process. To address these challenges, reconstructive surgeons must carefully assess each patient’s unique circumstances, select appropriate donor sites, and employ meticulous surgical techniques [3-5]. Furthermore, postoperative donor site dressings are vital for optimal healing and to minimize complications.
In this study, we introduce a novel approach for managing skin graft donor sites involving a combination of fenestrated polyurethane film and negative-pressure wound therapy (NPWT). This study aimed to compare the efficacy of this innovative method with that of conventional moist dressings using polyurethane foam. Parameters such as postoperative pain, time required for complete epithelization, and costs associated with different dressing approaches were evaluated.
MethodsFrom January 2021 to December 2022, STSG donor sites were managed with either a conventional method or one new method, without specifying indications for either method. A retrospective review was performed on 69 patients who received STSG, 35 of which were excluded for lacking complete documentation of donor site epithelization with clinical photography. Documentation of postoperative pain on days 1, 2, 3, and 7 after surgery was also considered; 11 patients with incomplete documentation for pain on such days were excluded. Among the remaining 23 enrolled, patients who were administered the new method on their donor sites were classified as group A (n=13), while patients who received traditional moist dressing on their donor sites were classified as group B (n=10) (Fig. 1).
The skin grafts were harvested in a standard manner, using an air-powered dermatome (Zimmer, Zimmer Surgical Inc.) adjusted to a thickness of 0.3 mm. In group A, a polyurethane film (Opsite Flexifix, Smith and Nephew Healthcare) fenestrated with a 20-gauge needle was carefully applied to the donor site of the skin graft. A hydrocolloid film (Comfeel, Coloplast) was carefully positioned along the periphery to protect the surrounding healthy skin from maceration. Finally, the NPWT system (CuraSys, CGBio Inc.) was affixed over the fenestrated film at a pressure of 100 mmHg in continuous mode for 14 days (Fig. 2). The conventional NPWT system was replaced with a portable NPWT (PICO system, Smith and Nephew Healthcare) if the patient was discharged from the hospital (Fig. 3). On the 14th postoperative day, both the film and NPWT were removed. The epithelialization rate of the donor sites was assessed, and in instances of incomplete epithelialization, traditional moist dressings were applied until completion. In group B, patients received the traditional moist dressing regimen; topical application of mupirocin ointment and polyurethane foam dressing (EasyFoam, CGBio Inc.) after STSG harvest, which was continued until complete epithelialization was achieved.
This study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Institutional Review Board of the Catholic University of Korea (IRB No. HC23RASI0084). Written informed consent was obtained from the patients to publish this paper.
PainPostoperative pain was evaluated using a numerical rating scale (NRS) in which patients were asked to pick a number between 0 (no pain) and 10 (worst pain). The NRS scores were recorded on postoperative days 1, 2, 3, and 7.
Reepithelization rateThe reepithelialization rate was defined as the number of days required for complete epithelialization of the donor site, with no additional dressings required. The decision was made by a single senior surgeon (J.H.L.) through clinical examinations and review of clinical photographs.
Treatment costCost comparisons were performed based on the cost of dressing materials and staff labor f (i.e., service cost). The treatment cost was standardized by dividing the total cost required for complete epithelization by the surface area in square centimeters.
Statistical analysisAll statistical analyses were performed using SPSS software (IBM Corp.). Frequency data are presented as percentages, and numerical data are presented as mean±standard deviation. Fisher exact tests were performed to assess categorical variables. Student t-test and Mann-Whitney test were performed to assess the continuous variables. Differences between the moist dressing group and the polyurethane film and NPWT dressing groups in terms of demographics and clinical outcomes were compared. P-value <0.05 indicated statistical significance.
ResultsOf the 23 patients included in the study, 13 received fenestrated polyurethane film and NPWT dressings (group A), and 10 received conventional moist dressings (group B) (Table 1). The mean ages of group A and group B were 65.5±11.7 years and 61.8±15.3 years (P=0.532), respectively. The mean surface area of STSG donor sites in group A and group B were 69.22±73.37 cm2 and 25.69±20.48 cm2 (P=0.136), respectively.
CostThe mean dressing cost per wound area (cm2) was significantly lower in group A (6,517±6,162 KRW [4.82±4.56 USD] vs. 23,036±27,157 KRW [17.05±20.11 USD], P=0.006) (Table 2).
DiscussionThe ideal dressing method for a STSG donor site should expedite the healing process, maintain appropriate humidity levels, absorb excessive exudate, reduce infection risks, ensure comfort, and provide a pain-free experience [6]. Common classifications of STSG donor site dressings include moist and non-moist categories. Moist dressings can be further subdivided into occlusive types (e.g., hydrocolloids and hydrogels) and semi-occlusive varieties (e.g., polyurethane films). Despite numerous studies addressing the management of STSG donor sites, a consensus on the optimal dressing method remains unclear.
In a national survey conducted by Geary and Tiernan in 2006 [7], alginate was selected as the most preferred dressing type for skin graft donor sites. An outcome study undertaken by Voineskos et al. in 2009 [8] demonstrated a leaning towards moist dressings. The most recent study, reported in 2018, involved a meta-analysis and systematic review of STSG donor sites [9]. This study concluded that, while lacking standardization, most dressings exhibited superior outcomes in terms of pain control and wound healing. Therefore, the authors presumed that moist dressings, particularly foam dressings, could be considered the conventional standard.
The use of a polyurethane film at the donor site was first introduced in 1975 [10]. This approach has gained support owing to its ability to maintain a moist environment through fluid retention, protect the wound surface from external shearing forces, its cost-effectiveness, and visual transparency, which allows for easy assessment. To address the shortcomings of film dressings, such as perforation, and excessive discharge leading to dressing instability or contamination, absorbent secondary dressings are used [11]. A prospective, randomized, double-blind clinical trial involving 50 patients revealed that polyurethane film dressing yielded superior results to those of Aquacel in terms of reepithelization rate and donor site pain [12]. Despite the excellent clinical outcomes associated with the use of polyurethane films, secondary dressings are still required to manage discharge.
Alternatively, the application of NPWT for managing STSG donor sites is recommended. Genecov et al. [13] reported that NPWT applied to petroleum gauze resulted in a higher epithelization rate than polyurethane film dressings. Similarly, Mitchell et al. [14] reported that the PICO portable NPWT device resulted in complete epithelialization of the STSG donor site in 10 patients within 2 weeks. Additionally, they asserted that single-use NPWTs provide enhanced convenience and portability.
We combined perforated polyurethane film dressings with NPWT to counteract the inherent drawbacks of conventional dressing methods. Although moist dressings (i.e., polyurethane foam) provide superior clinical outcomes, including faster epithelization and painless dressing changes, excessive discharge during the early postoperative period requires frequent change of dressings [12,15]. A study by Lee et al. [6] reported that the amount of discharge was the highest on the first postoperative day and decreased continuously afterward. By applying NPWT to the fenestrated polyurethane film, the exudate from the donor site was effectively controlled without changing the secondary dressing. In addition, the negative pressure contributes to the maintenance of the film over the defect area. Based on our study, the mean epithelialization time for the new type of dressing was 18.07±8.43. Conversely, patients requiring admissions extending beyond 2 weeks usually require no donor site dressing changes; however, for patients requiring hospitalization for less than 2 weeks, a shift to a portable NPWT (PICO system) was sufficient.
In group A, patients who were discharged with the PICO system returned to the outpatient clinic on the 14th postoperative day for the removal of the portable NPWT. For those who had not achieved complete epithelization, the next visit occurred between three to 10 day later, depending on the patient’s circumstances. Only one patient reported true delayed wound healing. The mean time for complete epithelization was lower in group A; however, the standard deviation was higher due to both the small sample size and variance in follow-up intervals. In future research, standardizing follow-up intervals for outpatient visits and increasing sample size are warranted.
Differences in baseline patient characteristics between the two groups were considered insignificant. Although the mean donor site surface area was larger in group A, the discrepancy was due to the broader range of defects in group A (range, 9–280 cm2) than in group B (range, 2–68.7 cm2). As no postoperative donor site dressings were necessary, both postoperative pain and dressing cost per cm² were significantly lower in group A.
The limitation of this study is its study design. The study was performed in a retrospective manner resulting in a relatively small sample size. The second limitation is inconsistency in pressure maintenance. The PICO system is known to deliver 80 mmHg of sub-atmospheric pressure, which differs from initial setting of 100 mmHg in CuraSys. In addition, the mean duration of conventional NPWT (CuraSys) application was 8.5 days ranging from 6 days to 14 days, whereas the mean duration of portable NPWT (PICO) was 5.5 days. Further investigations will be conducted in a prospective manner with randomization, consistent pressure maintenance with single device and standardized outpatient follow-up intervals.
Emerging dressing materials, such as bioelectric dressings, biological dressings, and dermal substitutes, have been developed and introduced in the market [16-18]. However, wider adoption is hindered by high costs and limited accessibility [19].
The combination of a fenestrated polyurethane film and NPWT resulted in reduced postoperative pain, faster reepithelization rates, and lower dressing costs than conventional moist dressings. The authors believe that this method can be considered an optimal choice within the armamentarium for managing STSG donor sites.
Fig. 1.Patient selection flowchart. A retrospective review of 69 patients who received split-thickness skin grafts from January 2021 to December 2023 was performed. After exclusion of 46 patients with lack of complete documentation, 13 patients (group A) who received fenestrated polyurethane film and negative-pressure wound therapy (NPWT), and 10 patients (group B) who received topical application of mupirocin ointment and conventional foam dressing (EasyFoam, CGBio Inc.), were compared. 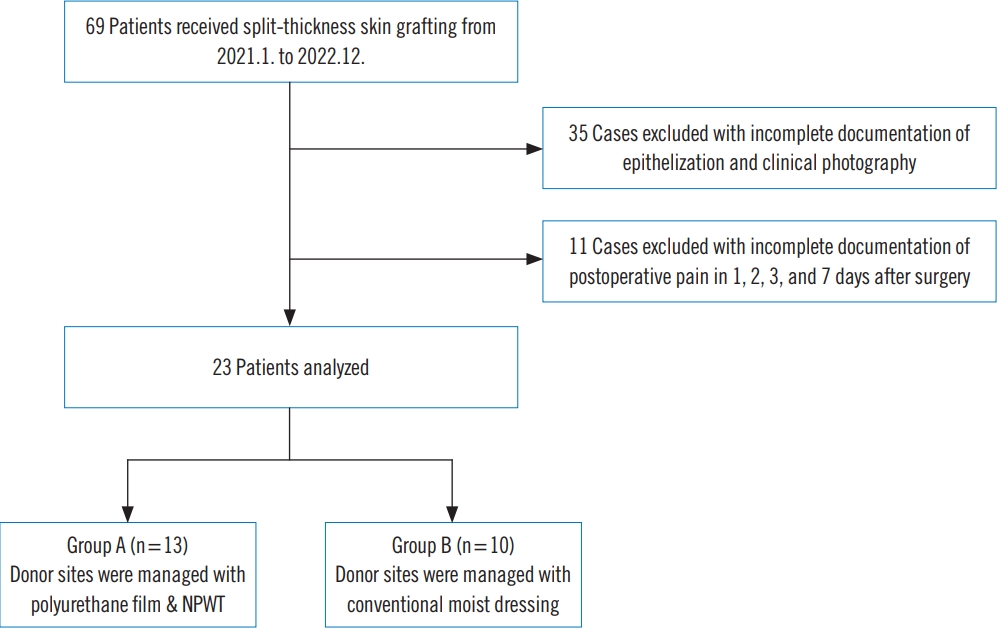
Fig. 2.Applying fenestrated polyurethane film and negative-pressure wound therapy (NPWT). Procedure for applying fenestrated polyurethane film and NPWT. (A) The size of the film is determined by the blood stain. (B) Multiple fenestrations were made with a 20-gauge needle. (C) A hydrocolloid dressing was positioned along the periphery to safeguard the surrounding healthy skin from maceration. (D) Finally, the NPWT system (CuraSys; CGBio Inc.) was applied over the fenestrated film at a pressure of 100 mmHg in continuous mode for 14 days. 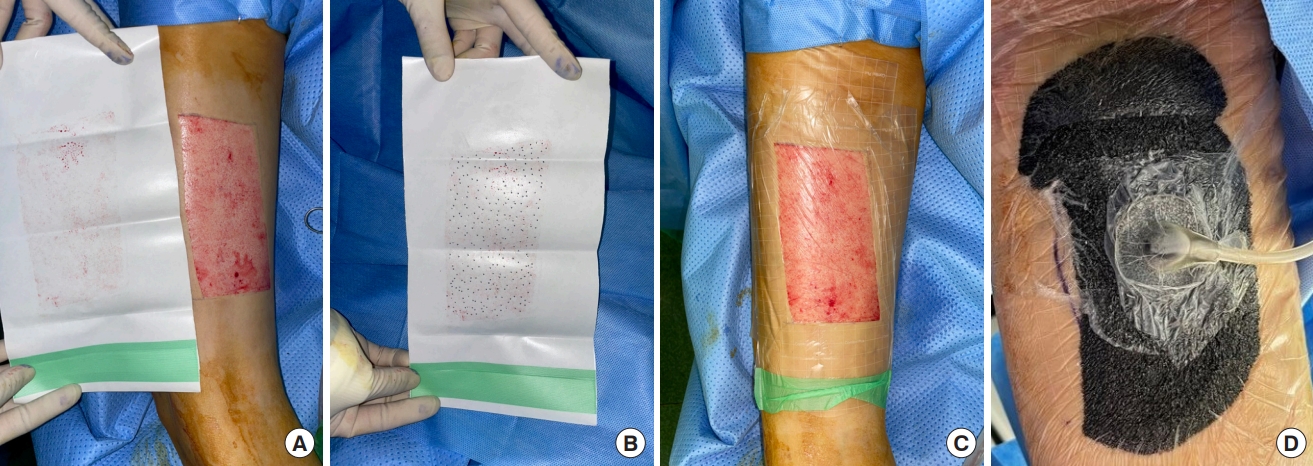
Fig. 3.Application of portable negative-pressure wound therapy (NPWT) device. In case of early discharge (i.e., <14 days), the conventional NPWT system was substituted with the portable NPWT system (PICO, Smith and Nephew Healthcare). 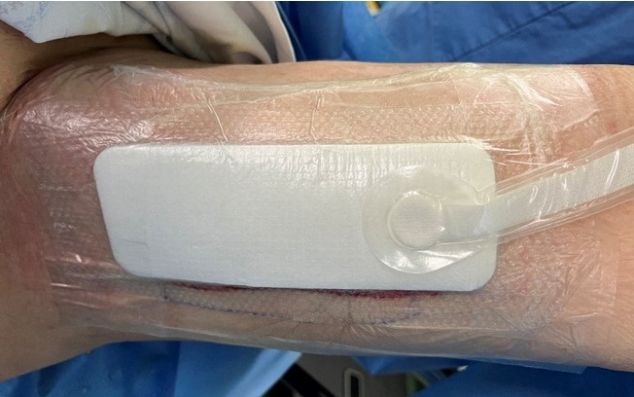
Fig. 4.Course of postoperative pain. Combined fenestrated film and negative-pressure wound therapy treated group (A) revealed a consistently lower mean pain score on all measured days than that of conventional moist dressing group (B). NRS, numerical rating scale; POD, postoperative day. 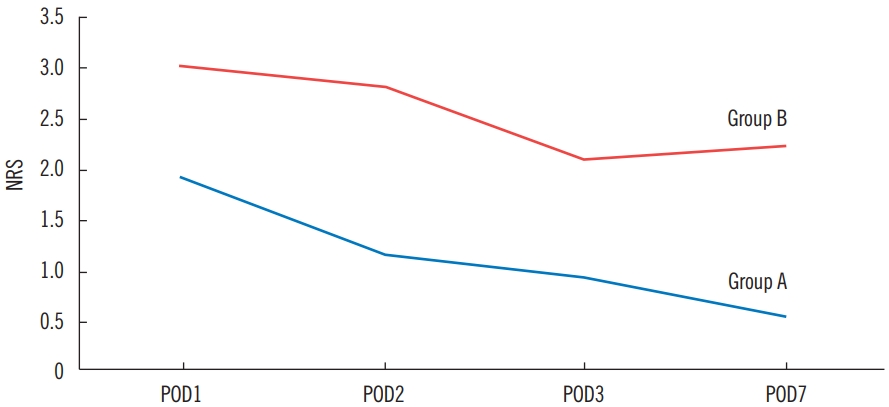
Fig. 5.Wound status after 2 weeks. This figure illustrates the wound status immediately after skin harvest (left) and after 2 weeks (right). Complete epithelization was achieved on postoperative day 14 in a 62-year-old man (A) and 88-year-old man (B). 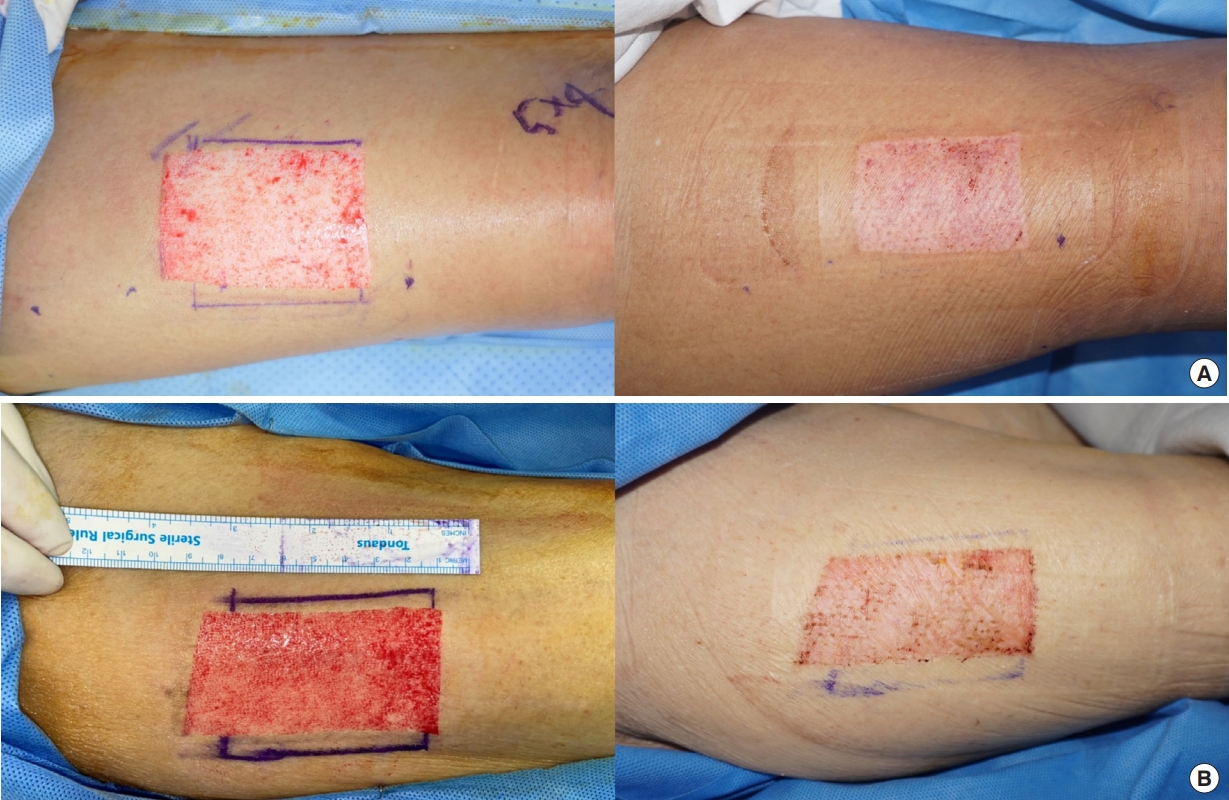
Table 1.Patient characteristics Values are presented as number (%) or mean±standard deviation. NPWT, negative-pressure wound therapy. P-values were determined by Student t-test (age) and Mann-Whitney test (area of donor site) for continuous variable and Fisher exact test for categorical variables (sex, diabetes, smoking history) P<0.05 denotes statistical significance. Table 2.Perioperative descriptive data
References1. Braza ME, Fahrenkopf MP. Split-thickness skin grafts. In: StatPearls [Internet]. StatPearls Publishing; 2023. Available from: https://pubmed.ncbi.nlm.nih.gov/31855388/.
2. Asuku M, Yu TC, Yan Q, et al. Split-thickness skin graft donor-site morbidity: a systematic literature review. Burns 2021;47:1525-46.
3. Akan M, Yildirim S, Misirlioglu A, et al. An alternative method to minimize pain in the split-thickness skin graft donor site. Plast Reconstr Surg 2003;111:2243-9.
4. Hallock GG. The cosmetic split-thickness skin graft donor site. Plast Reconstr Surg 1999;104:2286-8.
5. Sinha S, Schreiner AJ, Biernaskie J, et al. Treating pain on skin graft donor sites: review and clinical recommendations. J Trauma Acute Care Surg 2017;83:954-64.
6. Lee JH, Kang CS, Kim TG, et al. Patterns in the amount of discharge at split-thickness skin graft donor sites. Arch Aesthetic Plast Surg 2019;25:22-6.
7. Geary PM, Tiernan E. Management of split skin graft donor sites-results of a national survey. J Plast Reconstr Aesthet Surg 2009;62:1677-83.
8. Voineskos SH, Ayeni OA, McKnight L, et al. Systematic review of skin graft donor-site dressings. Plast Reconstr Surg 2009;124:298-306.
9. Serebrakian AT, Pickrell BB, Varon DE, et al. Meta-analysis and systematic review of skin graft donor-site dressings with future guidelines. Plast Reconstr Surg Glob Open 2018;6:e1928.
10. Birdsell DC, Hein KS, Lindsay RL. The theoretically ideal donor site dressing. Ann Plast Surg 1979;2:535-7.
11. Dornseifer U, Fichter AM, Herter F, et al. The ideal splitthickness skin graft donor site dressing: rediscovery of polyurethane film. Ann Plast Surg 2009;63:198-200.
12. Dornseifer U, Lonic D, Gerstung TI, et al. The ideal splitthickness skin graft donor-site dressing: a clinical comparative trial of a modified polyurethane dressing and Aquacel. Plast Reconstr Surg 2011;128:918-24.
13. Genecov DG, Schneider AM, Morykwas MJ, et al. A controlled subatmospheric pressure dressing increases the rate of skin graft donor site reepithelialization. Ann Plast Surg 1998;40:219-25.
14. Mitchell SL, Ray E, Cordeiro PG. Miniaturized negative-pressure wound therapy for split-thickness skin graft donor sites. J Reconstr Microsurg Open 2018;3:e46-9.
15. Chiang SH, Xu X, Cheung DST, et al. Comparison between aquacel and aquacel foam dressing on split-thickness skin graft donor site. J Wound Care 2021;30(Sup12):S14-20.
16. Son JW, Koo DY, Han SK, et al. Bioelectric dressing on skin graft donor sites: a pilot clinical trial. J Wound Manag Res 2022;18:92-7.
17. Poinas A, Perrot P, Lorant J, et al. Cicafast: comparison of a biological dressing composed of fetal fibroblasts and keratinocytes on a split-thickness skin graft donor site versus a traditional dressing: a randomized controlled trial. Trials 2019;20:612.
|
|