 |
 |

|
| Aims and Scope |
| About the Journal |
| About the Society (KWMS) |
| Editorial Board |
| Open Access |
| Principles of Transparency and Best Practice |
| Editorial Office |
| Subscriptions |


|
| Aims and Scope |
| About the Journal |
| About the Society (KWMS) |
| Editorial Board |
| Open Access |
| Principles of Transparency and Best Practice |
| Editorial Office |
| Subscriptions |

AbstractNon-radiographic axial spondyloarthritis (axSpA) is a subtype of axSpA characterized by chronic low back pain and stiffness but without radiographic evidence of sacroiliitis or spinal changes. As is the case with axSpA, early initiation of nonsteroidal anti-inflammatory drugs, tumor necrosis factor inhibitor agents, or physical therapy are often recommended for patients with non-radiographic axSpA. While the clinical course of non-radiographic axSpA is less well-known than axSpA, we present a case of a patient with non-radiographic axSpA manifesting as a sterile abscess in the sternum and costal cartilage. In this case report, we describe the diagnostic process for this rare manifestation and reconstruction of the large chest wall defect using a pedicled transverse rectus abdominis musculocutaneous flap.
IntroductionAxial spondyloarthritis (axSpA) is a chronic inflammatory disorder that affects the joints and enthuses of the axial skeleton, including the sacroiliac joints and the spine. Symptoms include chronic low back pain and stiffness, which are often accompanied by peripheral joint involvement, enthesitis, and dactylitis.
The diagnosis of axSpA is based on the presence of sacroiliitis and at least one typical spondyloarthritis (SpA) feature, or a positive human leukocyte antigen B27 (HLA-B27) test result and at least two other SpA features in patients with chronic back pain and age at onset less than 45 years (Fig. 1). The recommended treatment for this condition is the early initiation of nonsteroidal anti-inflammatory drugs (NSAIDs), physical therapy, and exercise. Disease-modifying anti-rheumatic drugs are not effective, and the use of high-dose glucocorticoids is avoided because of the associated long-term morbidity. In Europe and the United States, tumor necrosis factor (TNF) inhibitors, which have been approved for the treatment of this condition, have been shown to be effective along with NSAIDs [1].
Non-radiographic axSpA (nr-axSpA) is a subtype of axSpA characterized by chronic low back pain and stiffness but without radiographic evidence of sacroiliitis or spinal changes. Previous studies have shown that patients with nr-axSpA experience pain and disability at levels similar to those with axSpA, and demonstrate similar treatment responses to TNF inhibitors, despite a higher frequency of the disease in women and a higher proportion of negative HLA-B27 test results. nr-axSpA is an active area of research and discussion [2].
Here, we report a case of nr-axSpA manifesting as a sternal abscess that was difficult to diagnose. This case highlights the rare manifestation of nr-axSpA and the importance of radical resection as the treatment of choice. The resulting anterior chest wall defect was reconstructed using a pedicled transverse rectus abdominis musculocutaneous (TRAM) flap. This study aimed to increase awareness of this rare manifestation and management of nr-axSpA. This patient provided written informed consent for publication of this case report.
CaseA 56-year-old woman was admitted because of pain in the thoracolumbar area that did not improve with NSAIDs. Multifocal patchy enhancement at the disco-vertebral junction of the entire thoracolumbar and lumbar spine was found on lumbar-spine magnetic resonance imaging, and focal osteolysis corresponding to the enhancing portion of magnetic resonance imaging was found on computed tomography (CT); otherwise, there were no abnormal findings in either sacroiliac joint. Results of the HLA-B27 test were negative. To rule out infectious spondylitis, bone biopsy and culture were performed; however, these results were negative. Therefore, the department of rheumatology diagnosed nr-axSpA. The patient’s symptoms improved after administration of the TNF inhibitor, etanercept.
After 3 months, the patient complained of persistent sternal pain and a palpable mass-like lesion in the area with tenderness. A biopsy was initially performed at the affected site, which revealed granulation tissue with acute inflammation. However, over a period of 2 months, the biopsy site failed to heal and there was continuous pus discharge, prompting a CT scan. The CT scan showed soft tissue infiltration of the xiphoid process and both 5th costosternal joints, as well as multifocal abscesses at both pairs of the 5th to 7th costal cartilages (Fig. 2).
Therefore, the thoracic surgery team performed multiple debridements and utilized negative pressure wound therapy for the multifocal abscesses identified on the CT scan. Intraoperatively, it was observed that inflammation of the 5th and 6th costal cartilages extended posteriorly from the osteocartilaginous margin on both sides.
After multiple debridements, a large defect measuring approximately 27×12 cm in size and 5 cm in depth was ultimately created in the sternal region (Fig. 3). Tissue biopsy revealed granulation tissue with acute and chronic inflammation. No growth of aerobic or anaerobic bacteria was observed in microbial cultures, except for one instance where Acinetobacter baumannii was identified. However, it was not considered to be a true pathogen. We administered empirical treatment with piperacillin/tazobactam and ceftriaxone for 1 week each due to the absence of significant results in the microbial cultures. However, the patient’s white blood cell (WBC) count and C-reactive protein (CRP) levels did not decrease, with WBC ranging from 12,100 to 16,600 cells/mm³ and CRP levels between 12.00 and 13.86 mg/L, and intraoperative findings did not show any signs of controlled inflammation. Consequently, we suspected aseptic chondritis in nr-axSpA after excluding infection. However, the abscess was uniquely multifocal, which made it difficult to completely rule out infection, and after consultation with the department of rheumatology, we withheld from increasing the immunosuppressant dosage. Through multiple debridements and wide resections, the patient’s WBC count and CRP level eventually decreased to 10,000 cells/mm3 and 6.07 mg/L, respectively, which led us to consider reconstruction.
Due to concerns regarding the patency of internal mammary vessels located near the injury zone, reconstruction was performed using a pedicled TRAM flap. After flap insetting, congestion was observed, which prompted the decision for venous supercharging. An additional incision was made above the defect to expose the right 2nd and 3rd rib cartilages. The 3rd rib cartilage was partially removed, exposing the internal mammary vein (IMV) in the space between the 2nd and 3rd ribs. The ligated deep inferior epigastric vein was anastomosed to the IMV. As a result, the congestion resolved, and the flap remained stable (Fig. 4).
On postoperative day 7, discharge from the drain placed in the lower part of the flap became turbid, and a recurrent abscess developed at the left costal cartilage. Several microbial cultures obtained from the site repeatedly showed negative results. Throughout the entire process, both before and after reconstruction, a total of 16 microbial cultures were conducted, with only one instance of A. baumannii being identified. All other cultures showed negative results. Despite the low probability of A. baumannii being a true pathogen, after consulting with the infectious disease department, it was decided to administer tigecycline as a precautionary measure. Tigecycline was administered for a total of 3 weeks, but no improvement was observed in laboratory results, including WBC and CRP levels, or in the clinical drainage of the abscess.
After identifying recurrent abscesses around the remnant costal cartilage through intraoperative findings (Fig. 5) and a follow-up CT scan, radical resection was performed through consultation with the thoracic surgery department. The previous flap was elevated, and radical resection included total excision of the bilateral 5th to 6th costal cartilage and the connected rib bones as well as partial excision of the 7th cartilage. Following surgery, the patient recovered well with no respiratory complications and there was no recurrence of abscesses during the 6-month follow-up period (Fig. 6).
DiscussionThis was a case where diagnosis and treatment were difficult. A clear abscess was confirmed by radiologic examination and clinical intraoperative findings, and 16 pus cultures were performed on different days, intraoperatively and at bedside, to identify the bacteria. However, all results but one were negative. It could be suspected that the cause of the abscess was autoimmune rather than infectious. As diagnosed by the department of rheumatology, sterile costochondritis was accompanied by nr-axSpA; intraoperative findings demonstrated symmetric and bilateral posterior propagation of inflammation from the osteocartilaginous margin, characteristic for autoimmune disease.
In this case, there were many considerations regarding the selection of an appropriate donor to address the large defect in the sternal region. Various types of flaps can be utilized in anterior chest wall reconstruction, including the pectoralis major musculocutaneous (PMMC) flap, latissimus dorsi musculocutaneous flap, TRAM flap, and vertical rectus abdominis musculocutaneous flap. Each flap has its own advantages and disadvantages. The PMMC flap is suitable for sternal reconstruction with minimal rib involvement, being robust, easy-to-harvest, and versatile. However, it may not provide sufficient coverage for large defects [3]. The latissimus dorsi musculocutaneous flap is large and versatile, offering excellent tissue bulk, but it can be technically challenging to use and may have a limited reach [4]. The TRAM flap is large with excellent tissue bulk and skin coverage, but it requires a long pedicle and may cause abdominal hernias or weakness [5]. The vertical rectus abdominis musculocutaneous flap is large and versatile with excellent tissue bulk and skin coverage. However, it can be technically challenging to use, requires a longer operating time, and shares similar drawbacks as the TRAM flap, such as the risk of abdominal hernias or weakness [6]. PMMC and latissimus dorsi musculocutaneous flaps are the most commonly-used flaps for chest wall reconstruction and are suitable for use in the lower neck and upper third area of the sternum. The lower two-thirds segments of the latissimus dorsi muscle, based on secondary pedicles originating from the paraspinal perforators, can be used for inferolateral or posterior chest wall defects. However, in this case, the TRAM flap was selected because it was not suitable to use pectoralis major muscle or the latissimus dorsi muscle due to the inferior-medial defect of the sternum, and the considerable size and depth of the defect.
The patency of the internal mammary artery was confirmed on CT scan, but because the vessel was located in the inflammatory region, there were risks of adhesion when using it as a recipient vessel for a free flap. For this reason, the pedicled TRAM flap was selected; however, intraoperatively, flap congestion was observed after insetting. This congestion was suspected to be due to the insufficiency of the superior epigastric vein. Therefore, venous supercharging was performed by anastomosis of the ipsilateral deep inferior epigastric vein with the IMV located proximal to the defect site in an end-to-end manner. Subsequently, the flap was decongested. The use of the superficial inferior epigastric vein for supercharging in free TRAM flaps has been studied extensively. Previous studies have shown that in such cases, the surgical revision rate and the probability of partial or total flap loss are lower than when this vein is not preserved [7,8]. Although the superficial inferior epigastric vein could not be preserved in this case, it was confirmed that the ipsilateral inferior epigastric vein could be a good option for supercharging in the pedicled TRAM flap.
Even after flap reconstruction, a recurrent local abscess was observed at the margin where the cartilage was removed. Transcutaneous needle aspiration and intravenous steroid injection were attempted, but there was no improvement. After additional surgical resection, no recurrence occurred during 6 months’ follow-up. Therefore, it can be assumed that radical resection alone is the treatment of choice.
Radical resection of costal cartilage without limitation should be avoided. According to previous studies, respiratory complications, such as reintubation, pneumonia, and tracheostomy, were associated with an increased number of ribs being resected. Therefore, when removing the costal cartilage, only the inflamed parts should be selected to minimize the number of cartilages removed and preserve pulmonary function [9].
As nr-axSpA is not a common clinical manifestation, it is difficult to encounter more similar cases. However, if costochondritis occurs in a patient with nr-axSpA and abscess is suspected, first, infection should be ruled out, and active radical resection should then be considered.
Fig. 1.Axial SpA classification criteria from the Assessment of SpondyloArthritis International Society. SpA, spondyloarthritis; CD, Crohn’s disease; NSAIDs, nonsteroidal anti-inflammatory drugs; HLA-B27, human leukocyte antigen B27; CRP, C-reactive protein. 
Fig. 2.Multifocal abscess on computed tomography scan. Computed tomography scan shows soft tissue infiltration around the xiphoid process and both 5th costosternal joints, along with multifocal abscesses at both 5th to 7th costal cartilages (red arrows). 
Fig. 3.Intraoperative massive defect. (A, B) A 27×12 cm large defect, 5 cm in depth, is observed in the sternal region after multiple debridements, with inflammation extending posteriorly from the osteocartilaginous margin on both sides of the 5th and 6th costal cartilages. 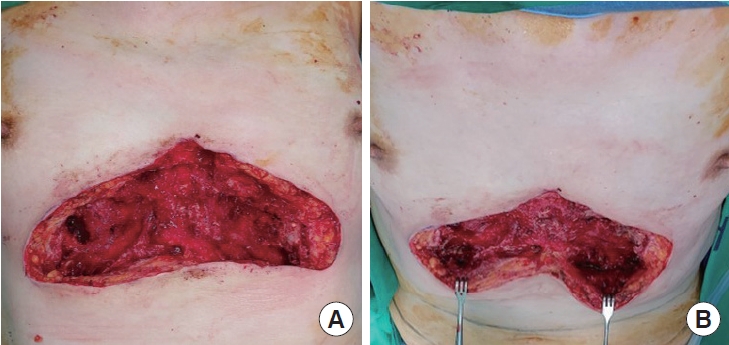
Fig. 4.Immediate postoperative photo. Congestion of the pedicled transverse rectus abdominis muscle flap is resolved after supercharging the ipsilateral deep inferior epigastric vein to the internal mammary vein. 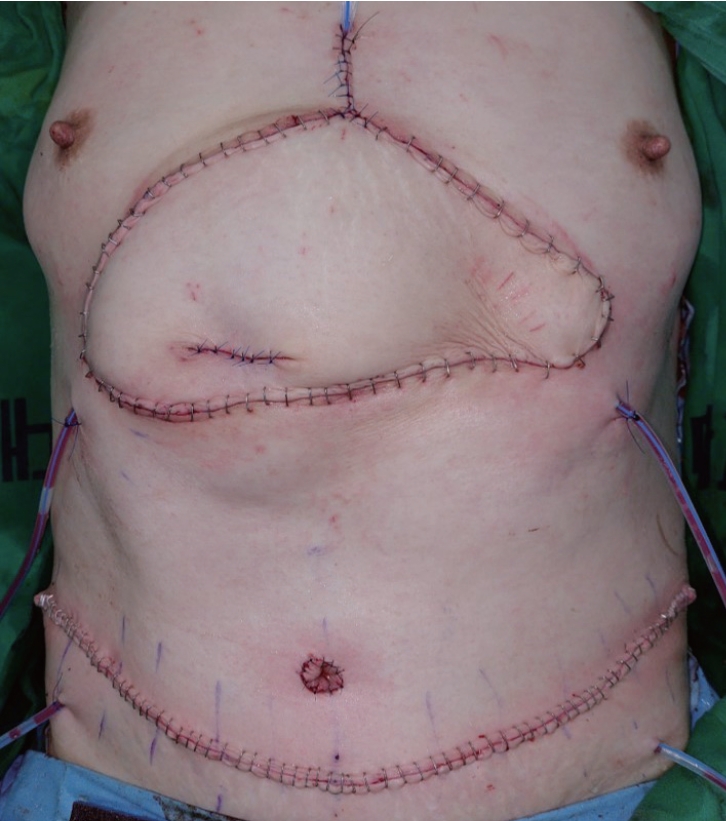
References1. Ritchlin C, Adamopoulos IE. Axial spondyloarthritis: new advances in diagnosis and management. BMJ 2021;372:m4447.
2. Glintborg B, Sorensen IJ, Ostergaard M, et al. Ankylosing spondylitis versus nonradiographic axial spondyloarthritis: comparison of tumor necrosis factor inhibitor effectiveness and effect of HLA-B27 status: an observational cohort study from the Nationwide DANBIO registry. J Rheumatol 2017;44:59-69.
3. Chen C, Gao Y, Zhao D, et al. Deep sternal wound infection and pectoralis major muscle flap reconstruction: a single-center 20-year retrospective study. Front Surg 2022;9:870044.
4. Vincent A, Hohman MH. Latissimus dorsi myocutaneous flap. In: StatPearls [Internet]. StatPearls Publishing; 2022. Available from: https://www.ncbi.nlm.nih.gov/books/NBK564377/.
5. Lin YN, Ou-Yang F, Hsieh MC, et al. Use of extended pedicled transverse rectus abdominis myocutaneous flap for extensive chest wall defect reconstruction after mastectomy for locally advanced breast cancer. Ann Plast Surg 2020;84(1S):S34-9.
6. Fujiwara M, Nakamura Y, Sano A, et al. Delayed vertical rectus abdominis myocutaneous flap for anterior chest wall reconstruction. Aesthetic Plast Surg 2006;30:120-4.
7. Razzano S, Figus A, Marongiu F, et al. Contralateral DIEV as an interpositional vein graft for venous supercharge in the salvage of a congested DIEP flap. microsurgery 2016;36:263-4.
|
|