Introduction
Venous ulcers are open skin wounds occurring in regions of the leg or foot affected by venous hypertension [1]. The pathophysiology of venous ulcers is a complex process with various associated signs such as varicose veins, chronic discharge, dermatitis, skin hyperpigmentation and fibrosis. These ulcers generally display a characteristic irregular shape with sharply demarcated borders, and are usually located in the peri-malleolar area [2]. Although ulcer depth is typically limited to the subcutaneous layers, infection can cause extensive deep tissue injury.
The prevalence of venous ulcers ranges from 0.7% to 2% of the general population [3–5]. They are the most common type of chronic leg ulcers and are three to four times more prevalent than arterial ulcers. Risk factors include old age, obesity, previous trauma history, immobility and genetic thrombophilic conditions such as factor V Leiden mutation [6]. Venous disease is more frequently observed in women [7,8] and pregnancy is known to increase the risk of developing a chronic venous ulcer [9].
Pathophysiology
Venous anatomy and physiology
An understanding of venous anatomy provides a better comprehension of chronic venous disease. The venous structure of the lower extremities can be categorized into superficial and deep systems. Superficial veins (e.g., greater saphenous vein, lesser saphenous vein and accessory saphenous vein) are located between the dermis and the muscle fascia, whereas deep veins (e.g., femoral vein, common femoral vein, deep femoral vein, popliteal vein, anterior and posterior tibial vein) are located beneath the muscle fascia [10,11]. These deep and superficial venous systems are connected by perforating veins. Most of the venous blood volume–approximately 85%–is reserved in the deep venous system.
Conductivity, capacitance, and directionality are the major components of venous physiology. Conductivity is related to low resistance in venous structures. This feature enables trans-portation of a large volume of blood without high energy ex-penditure. Capacitance refers to how easily the vein wall di-lates. Directionality is enabled by one-way valves in the venous structure generated from three physiological functions: respi-ration, cardiac contraction and locomotion. Venous dysfunction is induced from alteration of any of these three components.
Valve incompetence
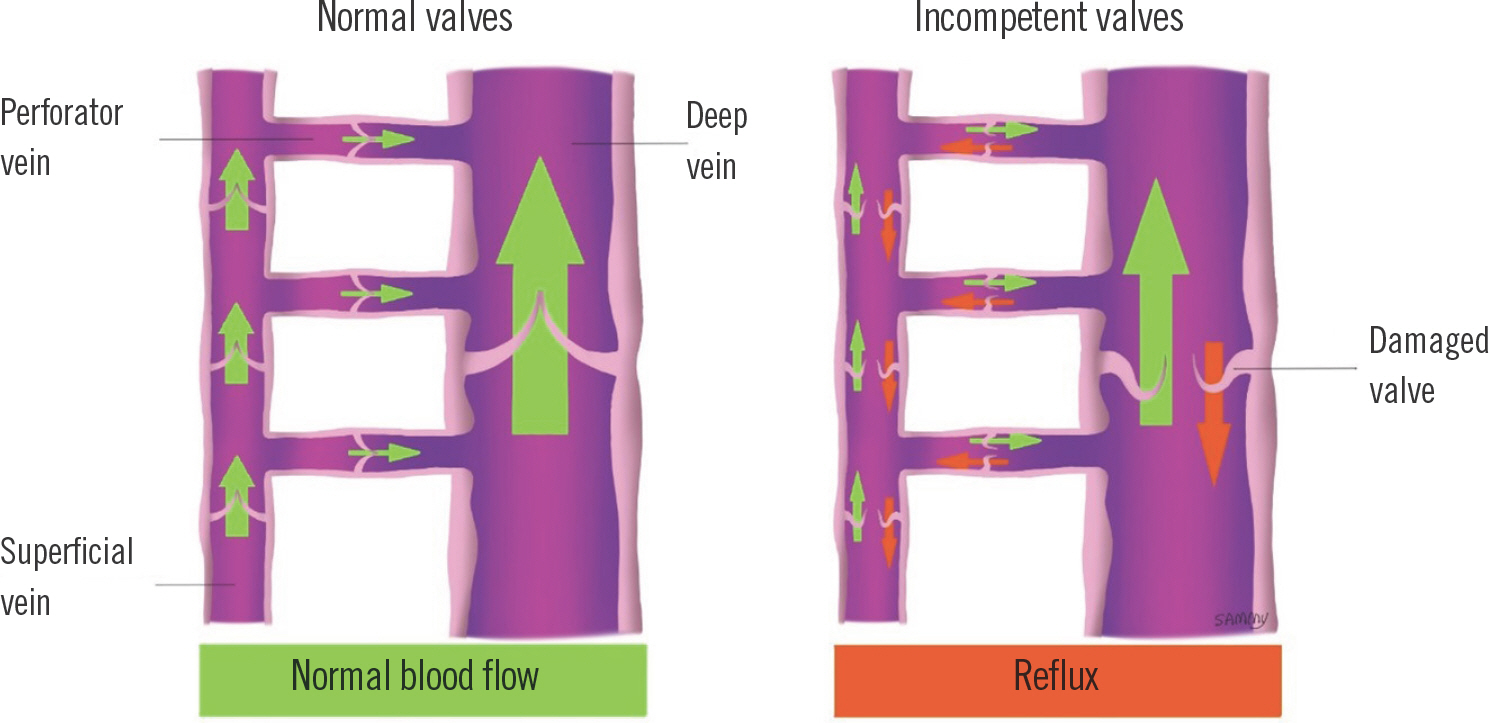
A constant unidirectional flow from the superficial into the deep venous system is ensured by competent valves in perforating veins. Superficial veins also must have competent valves to propagate flow into the more proximal deep veins. With incompetent valves, retrograde displacement of blood into the intramuscular veins and sinusoids can occur. This causes im-pediment of arterial inflow and decrease of muscle pump effectiveness (Fig. 1). Therefore, pump effectiveness and the pathways of blood flow direction are determined by the com-petence of venous valves.
Fig. 1.
Illustration of incompetent valves resulting in venous reflex. With competent valves, venous blood flows physiologically from the superficial system to the deep system through perforating veins (left). When damaged valves lose their function, reflux occurs which ultimately leads to venous hypertension (right).
Obstruction
Obstruction is an important factor in the pathogenesis of venous ulcers. Proximal venous obstruction has a higher prevalence in chronic venous disease and implies poorer outcomes in treatment [12]. Marston et al. [13] reported the importance of identifying venous obstruction in venous ulcer patients. Though frequently occurring in post-thrombotic veins, venous obstruction is difficult to detect non-invasively and thus often goes unnoticed. Intravascular ultrasound has proven its efficacy in assessment of the degree and extent of iliac vein ste-nosis compared to ascending phlebography which had previously been considered the standard [14].
Calf muscle pump
The theory of the calf muscle pump concept is a crucial com-ponent of venous physiology. Contraction of the calf muscles causes compression of the deep venous system, resulting in proximal propagation of blood. On the other hand, relaxation of the calf muscles causes expansion of the deep venous system followed by influx of blood from the superficial venous system through perforating veins. Dysfunctional calf muscles could be the prime cause for venous hypertension in some patients. Decreased ankle range of motion is also frequently observed in people with venous ulcers. Patients with chronic venous disease are prone to secondary lymphedema (phlebo-lymphedema) which is usually more severe at the ankle level. Because of restrictions in ankle motion, the effectiveness of the calf muscle pump decreases.
Genetic factors
Genetic mutation can be a predisposing factor for developing venous ulcers. Factor XIII is an important element for the for-mation of a fibrin matrix wound cover, further initiating a cascade of fibroblast growth and migration. Mutation in the F13A1 gene which encodes Factor XIII is known to be associated with increased size of venous leg ulcers and delayed wound healing [15]. A recent publication by Anwar et al. [16] summarized genetic mutations associated with poor healing or progression of venous ulcers.
Pathogenesis
Venous stasis theory
Introduced in 1917 by Homans [17], the term “postphlebitic syndrome” was used to explain the etiology of venous ulcer-ations. Based on previous literature, he described damaged valves and dilated walls as central elements in venous stasis. Homans hypothesized that hypoxia of the surrounding tissues is induced by this stasis, which ultimately leads to ulceration.
Arteriovenous fistula theory
Blalock [18] postulated the arteriovenous fistula theory from an analysis of hypoxia from chronic venous insufficiency. He compared blood samples from the femoral vein, greater saphenous vein, and varicose veins in patients with chronic venous insufficiency and samples taken from normal limbs. Higher oxygen concentration was noted in proximal veins of affected limbs. Therefore, he hypothesized that increased flow caused by arteriovenous fistulae rather than stasis was the cause of venous ulcers.
Fibrin cuff theory
Proposed by Burnand et al. [19], the fibrin cuff theory is based on a study of skin biopsies from patients with chronic venous disease. The biopsies revealed enlarged capillaries with a notable increase in pericapillary fibrin deposition. The investiga-tors hypothesized that venous hypertension caused microcir-culatory changes that enlarged the endothelial gap junctions, leading to extravasation of serous fluid which would include fibrinogen. The ensuing pericapillary fibrin cuffs would act as a barrier against skin nutrition and oxygenation.
White blood cell trap theory
Early activation of white blood cells resulting from venous hypertension has been demonstrated in previous studies. Coleridge Smith et al. [20] introduced a hypothesis in which white cell trapping results in neutrophil activation, consequentially dam-aging the surrounding tissues. Because white blood cells are known to be important mediators of ischemia and tissue dam-age, this theory is an essential part of the cascade of cellular and humoral events leading to venous ulceration.
Classification and diagnosis
Classification
The CEAP (Clinical-Etiological-Anatomical-Pathophysiologi-cal) classification is the current international standard for clas-sifying chronic venous disease [21,22]. The aspects of the classification are summarized in Table 1.
Table 1.
CEAP (Clinical-Etiological-Anatomical-Pathophysiological) classification for chronic venous disease
Diagnosis
The diagnosis of venous ulcers is clinically based on patient history and clinical presentation, augmented when necessary by diagnostic tests. While color-flow duplex ultrasound is currently the gold standard diagnostic procedure for chronic venous disease [10], other examinations can be performed in adjunct to duplex sonography.
The ankle-brachial index (ABI) is a non-invasive test which identifies peripheral arterial disease (PAD) in the lower extremity. If the ABI is less than 0.8, it is mandatory to evaluate and rule out arterial insufficiency because application of compression therapy in such instances can cause detrimental con-sequences.
Duplex ultrasound is a non-invasive examination which combines traditional ultrasound with Doppler ultrasonogra-phy for structural and functional evaluation of the venous system. Both arterial and venous blood flow can be visualized, with easy detection of obstructions and refluxes [23,24]. This also allows mapping of superficial, deep and perforating venous systems.
Photoplethysmography (also called light reflex rheography), also non-invasive, is a test which measures venous refill time. A probe is positioned on the skin just above the ankle, and the patient is instructed to perform brief periods of calf muscle pump exercises. Infrared light reflected by hemoglobin is mea-sured by the probe, assessing skin blood flow reduction caused by exercise [25]. The test measures calf muscle pump efficien-cy while also determining whether abnormal venous reflux is present.
Other imaging modalities such as computed tomography and magnetic resonance imaging can also be utilized. Although not routinely used for venous imaging, they can be used for evaluation of pelvic vein thrombosis due to the diagnostic limitations of duplex ultrasound in intrapelvic structures.
Treatment
The goal of venous ulcer treatment is to improve symptoms, prevent sequelae, and promote ulcer healing. Compression therapy and ancillary treatment (e.g., physical therapy, manual lymphatic drainage, and phlebotonics) constitute the back-bone of non-surgical venous ulcer treatment.
Compression therapy
The cornerstone of treatment for venous ulcers is medical compression therapy. While being both easy to apply and non-invasive, compression therapy effectively counteracts the mechanism of chronic venous disease (e.g., venous reflux and hypertension); therefore, the importance of compression can never be overstated. The following are examples of medical compression therapy: (1) short-stretch bandages, (2) long-stretch bandages, (3) Non-elastic bandages, (4) multicomponent systems, (5) ulcer stocking systems, and (6) adaptive compression systems.
Table 2.
Systems for compression therapy
Modified from Partsch. Phlebology 2014;29(1 suppl):140-5, with permission of SAGE Publications [26].
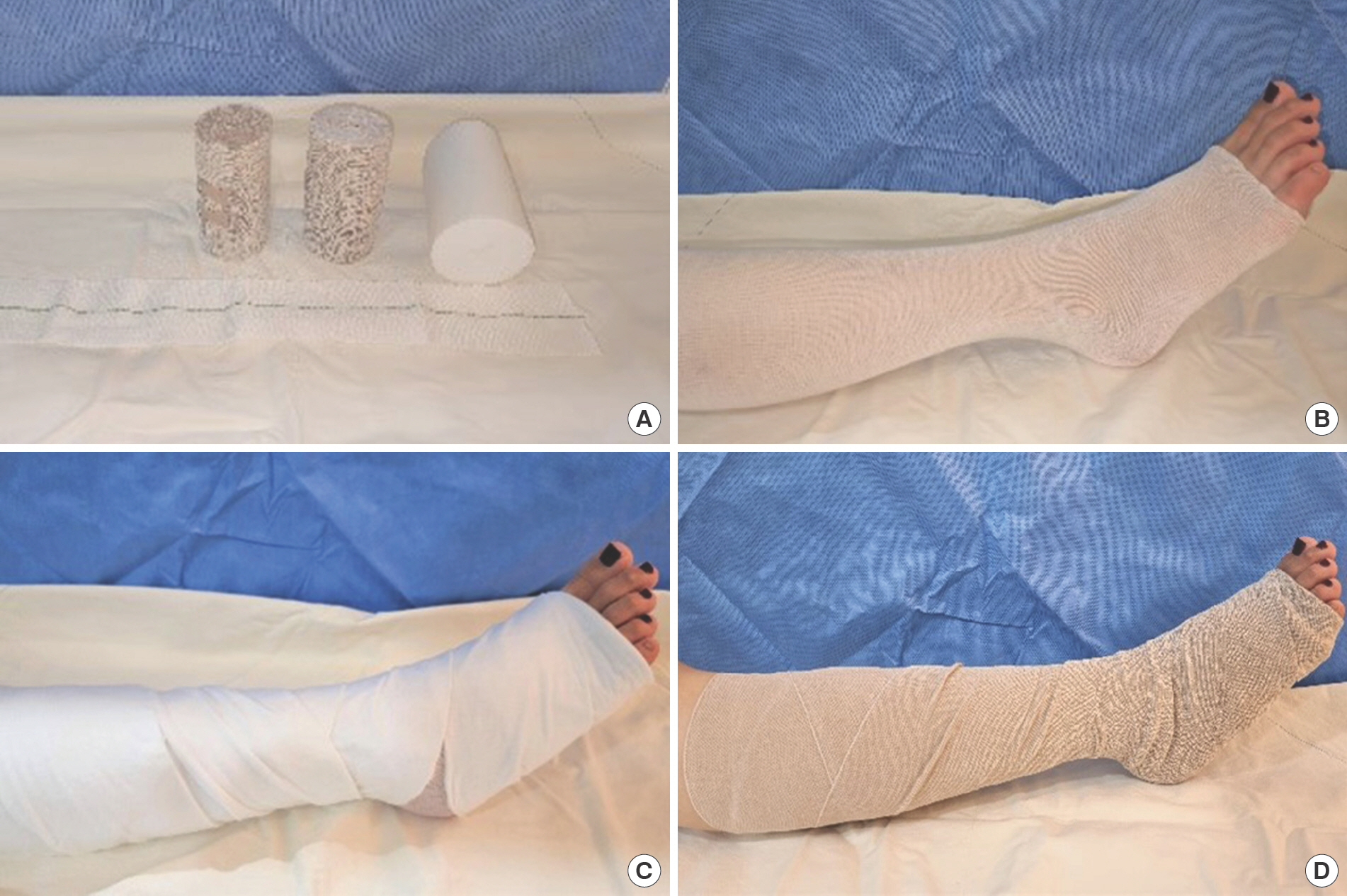
Short-stretch bandages have low elasticity (less than 100%). They are usually made of cotton and can provide high working and low resting pressures. A padding layer should preferably be used to prevent slippage (Fig. 2). The bandage should be renewed every morning.
Fig. 2.
Compression method using short-stretch bandages. (A) Materials, (B) tubular gauze, (C) underpadding, and (D) short-stretch bandage application with 50% overlap are displayed.
Long-stretch bandages are composed of cotton, polyamide, elastane and viscose. Because of their high elasticity (more than 100%), long-stretch bandages can provide low working and high resting pressures. As this can lead to pressure injury, using long-stretch bandages exclusively for strong compression is not recommended and overnight application should be avoided [27,28].
Non-elastic bandages such as zinc paste bandages (used in Unna boots) are notable for their very high working pressures and very low resting pressures. Their elasticity is below 10%. Zinc paste bandages consist of elastic fabric drenched in zinc solution or zinc paste. After wrapping the wet bandages around the affected extremity, compression pressure is devel-oped through solidification. The dried bandage becomes rigid and is maintained for several days [29,30].
Multicomponent systems commonly called “four-layer ban-dages” consist of different materials, including padding, compression, and fixation bandages. They provide great tolerability and a visual indicator assists in achieving optimal pressure [31]. Depending on the underlying wound status, these bandages may be maintained for up to 7 days, but more frequent change intervals are required in heavily exuding wounds.
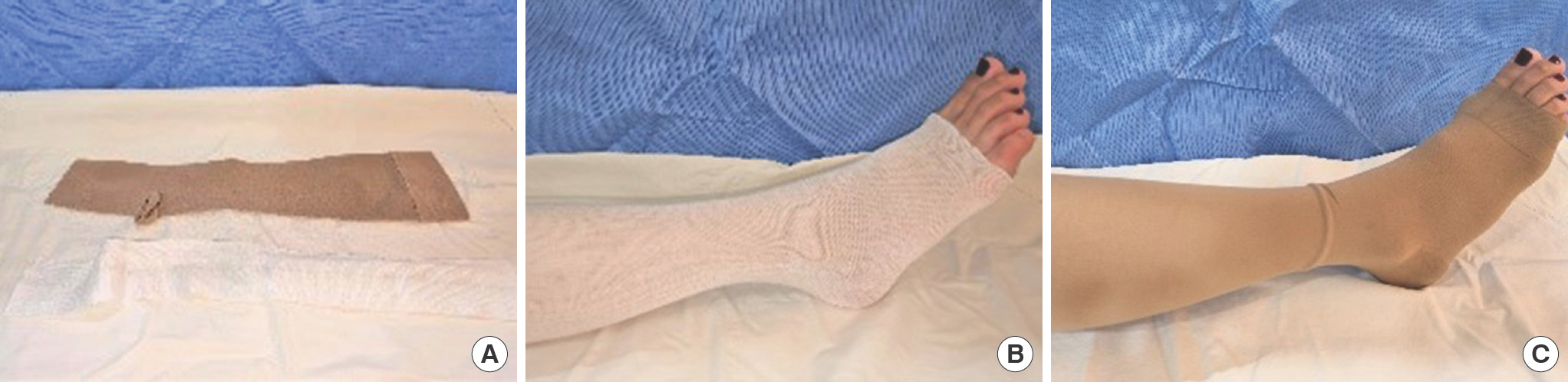
Ulcer stocking systems are usually made of two components: an understocking and an outer compression stocking. After training, many patients can readily apply them on their own (Fig. 3). The ulcer stocking system represents the treatment option of choice during the maintenance phase.
Fig. 3.
Compression method using ulcer stocking systems. (A) Material, (B) tubular gauze, and (C) ulcer stocking application are displayed.
Adaptive compression systems are also known as wrap systems or Velcro systems. These consist of short stretch material fixed by Velcro bands. Pressure can be adjusted at need to a comfortable level by the patient [32].
To properly understand the effects of compression therapy, one must know the distinction between resting and working pressure (Fig. 4). Resting pressure is defined as the pressure applied to the extremity at rest. This correlates to the strain exerted by the compression garment on relaxed muscles. Working pressure is a collaboration of both muscle contraction and external compression when the extremity is moving and muscles working. Therefore, the goal of compression therapy is to minimize the resting pressure and maximize the working pressure.
Fig. 4.
Illustration of comparison between resting and working pressure. Resting pressure refers to constant external pressure applied to skin and venous systems generated by the pressure garment (left). Working pressure refers to a dynamic pressure exerted on skin and venous systems generated from the combination of muscle contraction forces and the pressure garment (right).

Compression should be adjusted according to treatment stage, which can be divided into the decongestion phase and the maintenance phase. Edema control and promotion of ulcer healing are the primary aims in the decongestion phase. In this period strong compression methods, such as short-stretch bandages or multicomponent bandages, can be applied. Because of profuse edema and discharge, a combination of high absorbent wound dressings with daily renewal of bandages is generally recommended. Over time, decrease in exudate and congestion will allow twice-weekly bandage changes [28]. Completion of the decongestion phase leads to the maintenance phase. Compression during the maintenance phase can be successfully carried out by ulcer stocking systems [33]. However, protective padding should be applied to the edges of the tibia, the fibula head, and the ankle region with caution to prevent pressure injuries.
There is no clear evidence that proves superiority of a par-ticular type of compression. Regardless of the bandage type, it must be properly applied, which requires training [27]. Never-theless, studies suggest that high-pressure compression is more effective than low-pressure compression; and multiple layers of bandage may be necessary for adequate compression [34–36].
Some critical contraindications should be considered before applying compression therapy [27,28]. Absolute contraindications include advanced PAD, decompensated heart failure, and septic phlebitis. Relative contraindications include mild to moderate PAD, advanced peripheral polyneuropathy, chronic compensated heart failure, allergic reactions or intolerance to compression system materials, and treatment-related pain.
Physiotherapy
The goal of physiotherapy is to decrease venous pressure and edema by increasing calf muscle pump function through exercise or biomechanical stimulation. Lifestyle changes and spe-cial exercises can help in improving both calf muscle function and ankle mobility. Klyscz et al. [37] reported the benefits pro-duced by a venous rehabilitation program including four phases of training (warming, bike exercises, walking and relaxation). Another study that reviewed 16 papers concluded that increased physical activity, improved mobility, and foot exercises have a beneficial impact on lowering recurrence of venous ulcers [38].
Manual lymphatic drainage
Manual lymphatic drainage by massaging is a well-established therapy for lymphedema patients. This method increases both the transport capacity of the lymphatics and peripheral circu-lation [39]. One study reported that manual lymphatic drainage improved ankle joint mobility in venous ulcer patients with but did not improve wound healing [40]. Although there is currently a lack of supporting evidence, manual lymphatic drainage is a potential adjunctive treatment in some resistant ulcers.
Phlebotonics
Drugs such as natural flavonoids (e.g., rutoside, French mari-time pine bark extract, grape seed extract, etc.) and similar synthetic products (e.g., calcium dobesilate, naftazone, amin-aftone, chromocarbe) which are known to improve blood cir-culation are collectively referred to as venoactive drugs or phlebotonics. These agents are used to relieve symptoms such as pain, paresthesia, or restless leg but have no reported effect on ulcer healing [41]. Therefore, phlebotonics can be considered as an adjunct therapy to relieve related symptoms. It has been reported that a micronized purified flavonoid fraction (Daflon 500; Servier, Paris, France) reduces edema and related symptoms in chronic venous insufficiency [42]. A double blind placebo-controlled randomized study conducted in 2002 on coumarin and troxerutin (a flavonoid) revealed im-provement in edema control over placebo [43].
Novel therapies
Matrix metalloproteinases play a critical role in venous ulcer pathophysiology. Imbalance between proteinase activity and inhibition is a major cause of delayed wound healing in venous ulcers [44]. Due to the complexity of the molecular mechanism of venous ulcers, developing therapeutic targets has not been easy. A 2008 study demonstrated reduction of protease activity in venous ulcer exudate when a dressing product con-sisting of a combination of collagen and regenerated cellulose (Promogran; Johnson & Johnson, New Brunswick, NJ, USA) was applied [45]. The nano-oligosaccharide factor is a com-pound that promotes wound healing by inhibiting matrix me-talloproteinase activity. A randomized trial including 117 patients was conducted to compare a nano-oligosaccharide fac-tor treatment and Promogran treatment group [46]. The study concluded a promising effect of nano-oligosaccharide factor in treating chronic venous ulcers.
Conclusion
Venous ulcers are a common condition caused by venous reflux and obstruction ultimately resulting in venous hypertension. Numerous theories have been proposed to explain the pathogenesis of venous hypertension causing ulceration from structural to molecular levels. Based on CEAP classification, venous ulcers are clinically diagnosed, but color-flow duplex ultrasound remains the gold standard of all diagnostic modalities. Although a wide range of therapeutic options exist, compression therapy is the fundamental principle that can be applied in all disease stages. Adjunct therapies such as physiotherapy, manual lymphatic drainage, and phlebotonics can be considered to provide symptomatic relief.















