Introduction
Blast injuries have been considered a problem unique to military practice or warfare. However, terrorism and remnants of prolonged warfare have increased the frequency of explosions impacting civilians [1]. Accidental blast injuries to civilians have also been reported in relatively safe areas, such as South Korea, where misplaced landmines near the Korean War armistice line have exploded [2]. The detonation of a bomb or landmine near the chest wall can result in life-threatening internal injuries to vital organs in the thoracic cavity and external soft tissue injuries [3]. After primary management of the injured vital organs, the treatment of deep and blunt penetration wounds resulting from contaminated shrapnel is also critical. To prevent postoperative major and minor complications, large and deep soft tissue wounds of the chest wall resulting from an explosive injury and shrapnel usually require a multidisciplinary surgical approach including extensive debridement and a proper reconstruction plan [4,5]. This case presents chest wall defect reconstruction using a skin-paddled vertical rectus abdominis muscle (VRAM) flap and successful salvage of a delayed sternal wound infection in a rare civilian blast-related injury from a stray landmine in South Korea. Written informed consent was obtained from the patient.
Case
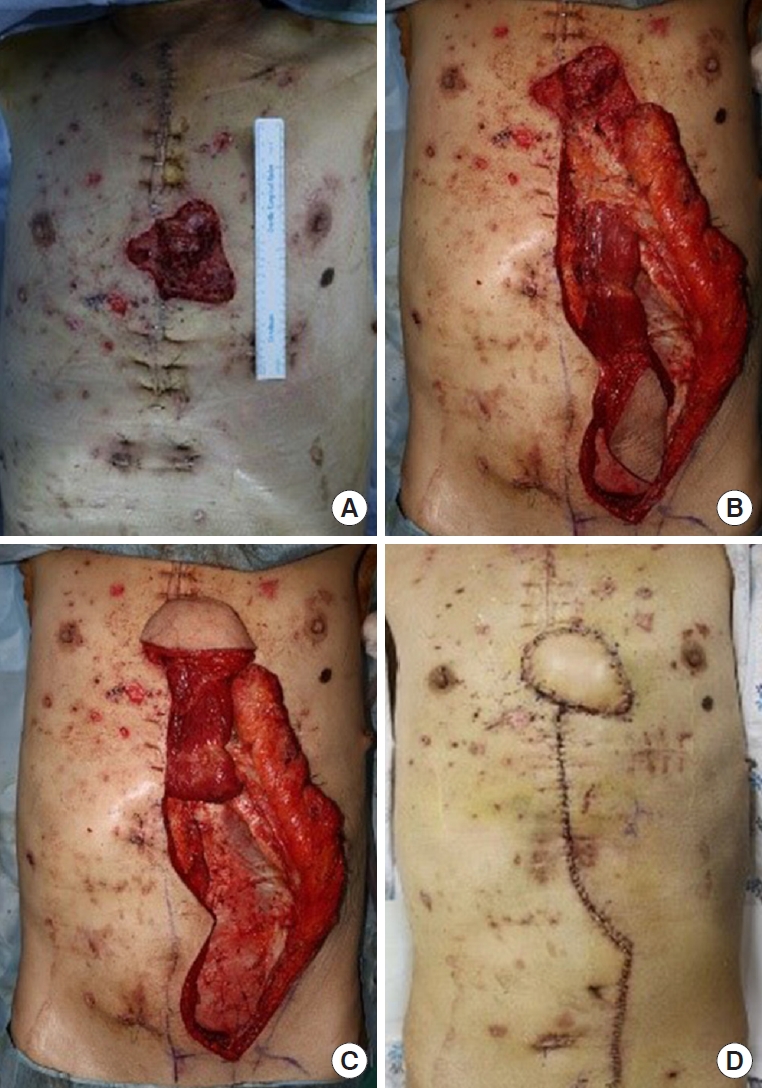
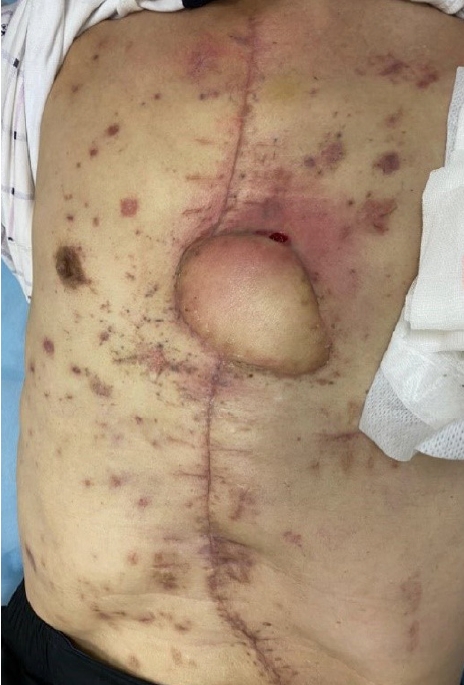
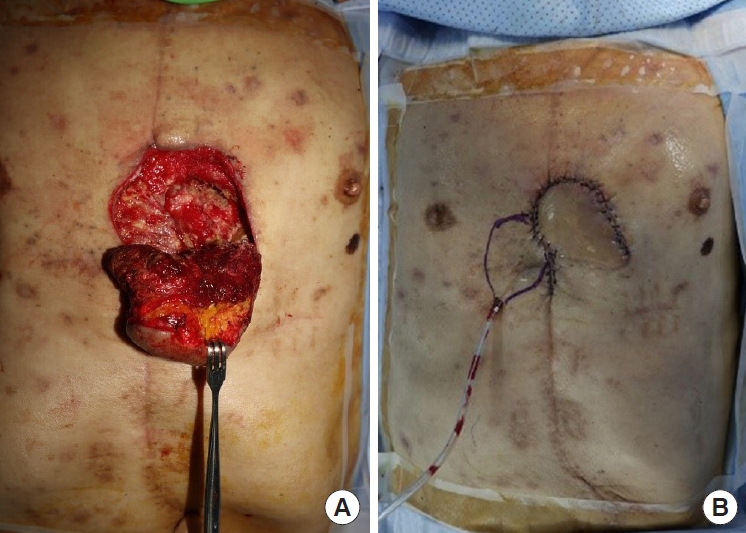
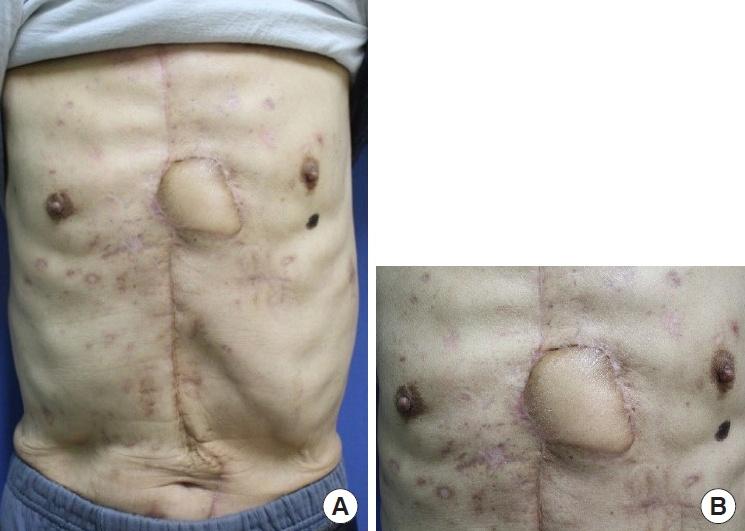
A 71-year-old man without underlying diseases was admitted to the hospital emergency room after an unknown explosive device detonated while he was attempting to sit on a fishing chair on a riverside. The debris from the device caused multiple injuries to his face, anterior thoracoabdominal region (Fig. 1A), and upper and lower extremities. On admission, his vital signs were unstable because multiple penetrating pieces of shrapnel had caused internal organ injuries, including hemopericardium and hemothorax. A thoracic surgeon performed emergency surgery via median sternotomy to explore the lungs and heart and remove the penetrated shrapnel from the pericardium. However, postoperative radiologic examination revealed shrapnel debris at multiple sites on the abdomen and chest wall. Foreign body removal followed by soft tissue debridement was performed, and wound cultures were obtained two more times. The wound bed was clean after several surgical debridements and serial wound cultures showed no bacterial growth. To prevent extensive wound infection, 4.5 g of piperacillin/tazobactam and 1 g of vancomycin hydrochloride were administered intravenously three times and twice a day, respectively, for the first 3 days. Under the advice of an infectious disease physician, prophylactic antibiotics were administered by switching to 1 g of fourth-generation cephalosporin three times a day for an additional 5 days. After the last debridement of the blast-related soft tissue injuries, soft tissue defects with sternal bone exposure measuring 1.5×2 cm and 5×4 cm remained on the lower third of the anterior chest wall (Fig. 1B). A soft tissue defect around the sternal notch also remained, but was repaired by bilateral advancement flap after the first debridement. Because of the widely contaminated and not fully demarcated wound on the lower third of the anterior chest wall, negative-pressure wound therapy (NPWT) was applied as a bridge between each debridement, and delayed chest wall reconstruction was planned. Three weeks after the initial trauma, no growth of any bacteria was found from final wound cultures. Therefore, the 7×7 cm skin and soft tissue defect were reconstructed with a superior epigastric artery pedicled VRAM flap (Fig. 2). One gram of a first-generation cephalosporin was administered prophylactically three times a day for 5 days after wound closure. The wound covered by the VRAM flap was stable, and the patient was discharged on the 10th day postoperatively. However, he visited our outpatient clinic 28 days postoperatively due to wound disruption with mild redness around the flap site and continuous purulent discharge (Fig. 3). The patient showed stable vital signs without systemic signs of infection. The laboratory results showed an increase in inflammatory markers with an erythrocyte sedimentation rate of 95 mm/hr, and a C-reactive protein level of 4.50 mg/dL, while the white blood cell (WBC) count was 8,580/μL and neutrophil count was 47% of the total WBC count. The growth of a fungus, Aspergillus terreus, was also revealed by fungal culture. Chest computed tomography showed sternal osteomyelitis, costochondritis, and mediastinitis. To salvage the previous flap, several surgical debridements were performed after re-elevating the previous VRAM flap through the upper half margin. The fifth and sixth rib cartilages and the adjacent half of the sternum were resected during radical debridement, and the stability of the chest wall framework was confirmed by a thoracic surgeon. NPWT was applied between serial surgical debridements, and 200 mg of voriconazole was administered intravenously twice a day followed by consecutive wound and fungal cultures. When the patient showed no further signs of clinical infection, and inflammatory markers with no growth of bacteria and fungus from serial cultures, the final defect was fully covered with local flaps by re-rotating the previous VRAM flap (Fig. 4). The wound was not disrupted again and the patient was discharged without complications 15 days postoperatively. The wound was also found to be stable without complications 3 months after the initial trauma (Fig. 5).
Discussion
The effects of blast injuries are classified as primary, secondary, tertiary, and quaternary [4,6]. Primary injuries are caused by pressure waves, whereas secondary injuries are caused by fragments of explosive devices and stone or soil debris scattered by the detonation [3,4,6,7]. Tertiary injuries are the result of bodily displacement, and while quaternary injuries include burns, inhalation of toxins, and exposure to radiation [4,6]. Since a pressure wave accelerates shrapnel contaminated with dirty soil debris in an uncontrolled direction, extensive penetrating injuries through vital organs such as the heart and lungs can occur after an explosion [6-8]. Therefore, the first priority should be resuscitation of the patient with adequate management of the vital organs. Once the vital signs are stabilized, meticulous wound management followed by soft tissue reconstruction is required. Any dirty fragments embedded throughout the body must be removed, and multiple minimal or radical debridements are required to prevent further wound infection [4]. Once the wound bed is cleaned, a reconstruction plan should be established according to the critical reconstruction cascade [4,5]. Pre-reconstruction NPWT has emerged as an effective method to replenish healthy granulation tissues and prevent infection [9,10].
Though appropriate reconstruction timing is set at 72 hours, delayed reconstruction is preferred for severely contaminated wounds [4]. Reconstruction of an extensive chest wall defect is challenging due to the anatomical and physiological complexity of the thoracic cavity [8,11]. Since inadequate reconstruction of an extensive chest wall defect results in prolonged ventilator dependence or significant respiratory compromise [5], skeletal stability with a prosthesis or a vascularized muscle flap is required for the successful closure of an extensive chest wall defect. The pectoralis major, latissimus dorsi, and pedicled rectus abdominis muscle flaps are considered the workhorse flaps used for various chest wall defects [9-12]. The pectoralis major muscle flap can be used primarily to cover defects in the upper chest or midsternum. The rectus abdominis myocutaneous or muscle flap can be used to cover defects in the lower pole of the chest. The latissimus dorsi muscle or musculocutaneous flap is also ideal for anterior and anterolateral chest wall defects. In our case, we planned delayed reconstruction in consideration of the severe contamination of the initial wounds after trauma on the anterior surface of chest wall and abdomen. After serial surgical debridements, the lower third anterior chest wall defect was confirmed to be clean with skeletal stability. Therefore, we decided to cover the defect using a VRAM flap because it is a well-vascularized soft tissue flap suitable for controlling infection and obliterating dead space [11,13]. It is also a versatile and robust muscle flap that yields a considerable volume of local tissue with similar texture and appearance to the chest region [11].
Open chest surgery through median sternotomy can be performed in victims of penetrating or severe blunt injuries involving the heart and lungs. However, wounds associated with blast shrapnel are vulnerable to infection due to the broad area of injury consisting of bomb remnants and contaminants such as debris and surrounding dust [4]. Deep sternal wound infections have also been reported to occur in approximately 1% to 5% of patients who undergo open chest surgery with median sternotomy [9]. Therefore, open chest surgery on a blast injury increases the risk of acute and delayed infection of both bones and soft tissues [9,10]. Delayed infection occurring 2 to 3 weeks postoperatively is referred to as a sternal wound type II, which is characterized by a positive wound culture with purulent mediastinitis, cellulitis, costochondritis, and osteomyelitis [9,14]. In cases of delayed infection, conventional approaches such as radical debridement and irrigation and drainage are attempted until the patient presents no systematic signs of infection and the wound has healthy granulation tissue with minimum exudate [9,10]. Several recent studies have also reported success after using NPWT as a bridge to control infection and fill the dead space with healthy granulation tissue before wound closure by flap surgery [9,10,14]. Delayed reconstruction can eventually be performed in the absence of serial wound cultures and residual infected bone and soft tissue by either elevation of a new flap or re-use of the previous flap.
Delayed fungal infections of the sternum after thoracic surgeries have also been rarely reported [4,15]. The majority of fungal infections occur in immunocompromised hosts, including patients with profound neutropenia, solid organ or stem cell transplant, and human immunodeficiency virus/acquired immunodeficiency syndrome [15]. Cases of Aspergillus species infections of the sternum and rib have been reported after cardiac surgery [15], and Mucormycetes infection on various body parts among victims of blast injuries has also been reported [4]. Complex blast injuries penetrated by shrapnel and aerosolized with environmental material are more susceptible to invasive fungal infection [4]. Because fungal infection progresses slowly and leads to disastrous results, infected wound biopsy and serial microbiological cultures are required for definite diagnosis. In the setting of culture-proven or strongly suspected fungal infection, several cases have been successfully treated with surgical debridement and administration of voriconazole [4,15]. In our case, we controlled the fungal infection by resecting the infected rib cartilage and sternum with intravenous administration of voriconazole and application of NPWT. The defect was successfully covered by the previous VRAM flap without requiring additional reconstructive surgery.
Many studies have reported on the mechanisms, pathophysiology, and treatment of various blast-related injuries on the chest wall after explosions such as bombs and landmines [1,3-8]. However, cases of chest wall reconstruction with delayed infection after blast-related injuries in the Korean population have not yet been reported in the literature. In our case, delayed reconstruction of the chest wall defect was performed after soft tissue infection control, following the treatment of vital organs and removal of foreign bodies. We reconstructed the lower third of the chest wall with the VRAM flap 22 days after the initial trauma, but signs of deep sternal wound infection, such as costochondritis and mediastinitis, occurred 20 days postoperatively. Wound disruption was managed by several radical debridements and NPWT application. After the infection subsided, the defect was covered by the previous flap and complete healing was achieved. Early chest wall reconstruction in blast injuries is preferred within 72 hours from the initial trauma; a limitation of this case is that the sternal wound infection may have been caused by delayed reconstruction. We hereby present the findings of a rare case of chest wall injury caused by an explosion in South Korea with successful chest wall reconstruction achieved despite delayed infection.