 |
 |

|
| Aims and Scope |
| About the Journal |
| About the Society (KWMS) |
| Editorial Board |
| Open Access |
| Principles of Transparency and Best Practice |
| Editorial Office |
| Subscriptions |


|
| Aims and Scope |
| About the Journal |
| About the Society (KWMS) |
| Editorial Board |
| Open Access |
| Principles of Transparency and Best Practice |
| Editorial Office |
| Subscriptions |

AbstractBackgroundNegative pressure wound therapy (NPWT) facilitates recovery of pressure injuries. This study aimed to investigate whether preoperative NPWT affected complications after musculocutaneous flap reconstruction of pressure injury wounds that required surgical treatment.
MethodsOf 71 patients with sacral pressure injury wounds, 28 did not undergo preoperative NPWT (no-NPWT group), while 43 received preoperative NPWT. Both groups received flap coverage for pressure injury wounds. Differences in complications such as wound dehiscence, hematoma, and seroma were compared between the two groups.
ResultsIn the no-NPWT group, seven postoperative complications occurred, of which five were wound dehiscence and two were seroma. There were no hematomas. In the NPWT group, a total of nine postoperative complications occurred, including two cases of wound dehiscence, six hematomas, and one seroma (P=0.029).
ConclusionThe overall incidence of complications was higher in the no-NPWT group. The incidence of hematoma was significantly higher in patients with preoperative NPWT. NPWT before reconstruction of pressure injury wounds may assist wound healing after the surgical treatment. However, care must be taken because NPWT may increase the risk of hematoma after surgical treatment.
IntroductionNegative pressure wound therapy (NPWT) is commonly applied to various wounds to aid recovery. In addition to its direct effects, including maintaining a moist wound environment and removing edema, NPWT has indirect effects, including reducing bacteria and increasing blood flow, collagen synthesis, and cytokine and growth factor levels associated with wound healing [1-4]. NPWT can thus aid wound recovery, even in wounds that are difficult to heal, through direct or indirect effects that are critical for recovery.
We applied NPWT to patients with sacral pressure injuries. Sacral pressure injuries are chronic wounds that do not heal easily, rarely recover fully with NPWT only, and whose recovery takes a relatively long time. Surgical reconstruction is often necessary for a complete repair.
Several complications occasionally occur after musculocutaneous flap coverage of pressure injury wounds. Typical postoperative complications include wound dehiscence, hematoma, and seroma (Fig. 1). These complications differ slightly based on whether NPWT was applied. Accordingly, we aimed to statistically analyze the postoperative complications of NPWT when applied to pressure injury wounds before reconstruction surgery. Although preoperative NPWT has been studied [5], there is a lack of information regarding its application in pressure injuries. In addition, no studies have identified differences in complications between patients who underwent preoperative NPWT and those who did not. Thus, we aimed to find more effective methods of applying NPWT and precautions required during its application to reduce complications after performing musculocutaneous flap coverage for pressure injury wounds.
MethodsThis study involved patients who underwent surgical reconstruction for pressure injury wounds between April 2018 and March 2020. We selected patients with sacral pressure injuries that were stage 3 or higher to reduce differences related to staging. The wounds were classified according to the 2019 National Pressure Injury Advisory Panel (NPIAP) [6]. All patients underwent surgical reconstruction for pressure injury wounds, which was performed by a plastic surgery specialist via the musculocutaneous V-Y advancement flap method (Fig. 2).
Patients who met the inclusion criteria for this study had a sacral pressure injury of stage 3 or more, hemoglobin A1C (HbA1C) <8 (if the patient had diabetes), and adequate nutrition status (indicated by a serum albumin level ≥2.0 g/dL). We excluded patients with uncontrolled infection wounds, i.e., patients who could not be confirmed to be free of bacteria in wound culture tests twice in a row at 1-week intervals; patients with an HbA1C ≥8 or uncontrolled diabetes; patients with hemoglobin <7 g/dL or hematocrit <20%; and malnourished, immunodeficient, or immunocompromised patients. In addition, patients at high risk of bleeding were excluded, including those with prothrombin time >15 seconds or activated partial thromboplastin time >50 seconds, and patients without adequate wound hemostasis. Patients who received anticoagulants or platelet aggregation inhibitors had to discontinue their intake of those medicines for at least 1 week before surgery.
Seventy-one patients (40 males and 31 females) were included in this study. Of these, 28 patients did not undergo prereconstruction NPWT; these individuals comprised the no-NPWT group. The 43 patients in the NPWT group underwent NPWT at least 8 days before reconstructive surgery.
Both groups underwent position change at 2-hour intervals during the preoperative and postoperative periods. The NPWT group was subjected to at least one surgical debridement procedure for removal of necrotic tissue, and the NPWT unit was replaced 2–3 times weekly. The pressures used during NPWT were 80–125 mmHg. The no-NPWT group also underwent serial surgical debridement using foam dressing and hydrofibers or topical antiseptic impregnated gauzes as dead space fillers. Before flap surgery, patients from both groups were subjected to bacterial culture tests at weekly intervals for wound infection control. The target antibiotics were administered for at least 2 weeks based on the culture test results. Postoperative clinical progress was tracked for a minimum of 3 months and a maximum of 2 years. We recorded all instances of postoperative wound dehiscence, hematoma, and seroma in both groups.
Fisher exact test was used to determine the incidence of complications between the two groups. Statistical analyses were conducted using SPSS version 27.0 (IBM Corp., Armonk, NY, USA). In addition, statistical evaluation was conducted to determine the relationship of the preoperative application of NPWT to pressure injury wounds with the occurrence of wound dehiscence, hematoma, and seroma. The study was approved by the Institutional Review Board of Pusan National University Yangsan Hospital (IRB No. 05-2021-204). The need for informed consent was waived because the study was retrospective in nature.
ResultsThe study cohort consisted of 71 patients, including 40 males and 31 females, with an average age of 60.3 years. Based on the 2019 NPIAP staging system, eight patients had stage 3 sacral pressure injuries. Of those, three underwent NPWT, while five did not. In contrast, 62 patients had stage 4 sacral pressure injuries. Of these, 40 patients underwent NPWT, while 22 did not. One patient had a deep tissue injury without NPWT applied. The average size of the sacral pressure injury wound was 61.1 cm2 in the no-NPWT group and 68.0 cm2 in the NPWT group. The overall average size of the sacral pressure injury wound of the two groups was 65.2 cm2. Preoperative NPWT applications ranged from 8 to 171 days, with an average of 33.7 days (Table 1).
The P-value for the association between the sex of patients and complications such as wound dehiscence, hematoma, and seroma was 0.065, which was statistically insignificant. The P-values for the patients’ ages, pressure injury stages, and preoperative size of the pressure injury were 0.289, 1.000, 0.783, respectively. However, the P-value for complications that were attributed to NPWT application before surgical reconstruction of pressure injury wounds was 0.029, which was statistically significant. There were seven cases of complications in the no-NPWT group, including wound dehiscence (n=5) and seroma (n=2). In the NPWT application group, there were nine cases of complications, including wound dehiscence (n=2), hematoma (n=6), and seroma (n=1). Among all the complications, wound dehiscence and seroma occurred slightly more often in the no-NPWT group. In contrast, only hematoma occurred more frequently in the NPWT group (n=6); no instances of hematoma were noted in the no-NPWT group. In addition, the average application period of preoperative NPWT was 7.4 days for patients with wound dehiscence, 17.7 days for patients with seroma, and 43.7 days for patients with hematoma. Therefore, the occurrence of hematoma was higher in patients who underwent NPWT application for a longer period of time (Table 2).
DiscussionNPWT was first used to manage soft tissue injuries in open-fracture wounds [7]. In recent years, wound recovery after the application of NPWT in animal models has facilitated the development and widespread use of various NPWT systems [8,9]. NPWT is a popular treatment modality that aids wound recovery in patients with both acute and chronic wounds [10]. NPWT is effective even in wounds difficult to treat with simple dressings such as necrotizing infections, diabetic foot ulcers, and pressure injuries.
NPWT assists in the management of complicated or dirty wounds, compared to traditional dressings which need to be changed frequently as they dampen with wound discharge over time, making exudate management difficult. In contrast, NPWT only requires unit changes every 2–5 days because the system immediately removes exudates from the wound surface via absorption. Since dressing changes are less frequent, NPWT can reduce pain from dressing changes. Conversely, the NPWT device can be cumbersome to manage and can potentially interfere with the patient’s mobility. Moreover, it cannot be applied to exposed vital structures, sites with vascular exposure, or malignant tissues [11,12]. The authors applied NPWT to sacral pressure injuries in consideration of these characteristics of NPWT. Application of NPWT to sacral pressure injury promotes wound recovery and helps maintain wound hygiene by minimizing potential sources of contamination. NPWT appears to be effective and efficient for wound management of pressure injuries.
The application of NPWT to pressure injury wounds does not necessarily result in full recovery. Even in cases where NPWT was applied to the pressure injury wound for a maximum of 171 days, flap surgery was eventually required. The average application period of NPWT to pressure injury was 33 days, followed by complete recovery of the pressure injury wound through surgical reconstruction. Application periods were extended because wound recovery decreases substantially following 1 month of NPWT application. Andersson in 2018 and Mari in 2019 demonstrated that the rate of wound shrinkage with NPWT gradually decreases over time [13,14].
Postsurgical complications, including wound dehiscence, hematoma, and seroma, can occur after flap coverage. Out of 71 cases in total, there were 16 cases of complications (22.5%). The complication rates were 25.0% in the no-NPWT group and 20.9% in the NPWT group; the rate being slightly lower in the latter group. However, wound dehiscence and seroma were more common in the no-NPWT group, while hematoma only occurred in the NPWT group. Furthermore, hematoma occurred more frequently in patients with a longer application period of preoperative NPWT. In particular, hematoma occurred in those with a minimum preoperative application period of 28 days and an average of 43 days. Thus, preoperative NPWT was significantly associated with hematoma after reconstructive surgery for pressure injuries (P=0.029). This may be due to increased wound perfusion and neovascularization in NPWT [1].
Complications that might occur after the surgical reconstruction of pressure injury wounds, particularly hematoma, can be influenced by various factors. For example, the duration of anticoagulant use, infection, osteomyelitis, posture, surgical site forces, and drainage tube removal timing can affect postoperative complications. We discontinued the use of anticoagulants at least 7 days before the surgery, managed preoperative wound infection using antibiotics, imposed limitations on supine and sitting positions after surgery, and strictly controlled contact with the surgical site. All drains were normally removed 1–2 weeks after the surgery. Incomplete control of all variables might be a limitation of this study. However, since only one out of the six cases of postoperative hematoma had a history of anticoagulant administration, the anticoagulant was not believed to have a huge impact on the study results.
In conclusion, the incidence of complications after reconstructive surgery for pressure injury was lower in the NPWT group than in the no-NPWT group. NPWT had positive effects on the preoperative treatment of pressure injuries. However, the incidence of hematoma was significantly higher when preoperative NPWT was applied for more than 1 month. This might be related to angiogenesis promoted by NPWT, which increased wound perfusion. Additional studies including a higher number of patients with a correlation between NPWT and hematoma are required. We found that NPWT can be helpful in effective and efficient wound management before the surgical reconstruction of sacral pressure injuries and may reduce postoperative complications. However, prolonged application of NPWT could increase the risk of postoperative hematoma. In addition, bleeding control should be prioritized during the surgery.
Conflict of InterestThis study was supported by a 2020 research grant from Pusan National University Yangsan Hospital. Otherwise, no potential conflict of interest relevant to this article was reported. Fig. 1.Typical complications after pressure injury surgery. (A) A patient with wound dehiscence. (B) The evacuation procedure in a patient with postoperative hematoma. (C) Aspiration conducted in a patient with seroma. 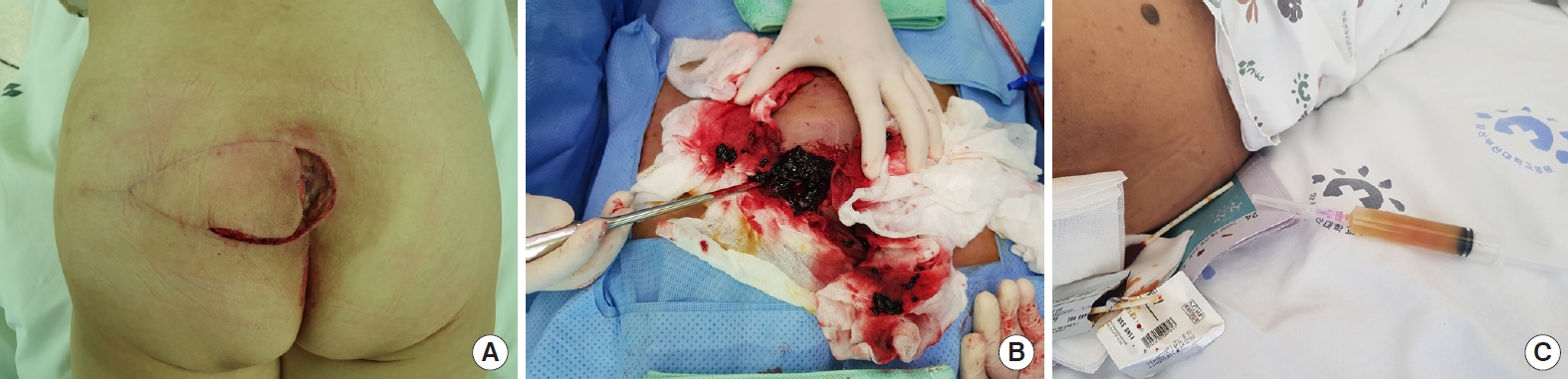
Fig. 2.A case of a 72-year-old male patient. (A) Application of negative pressure wound therapy. (B) Before surgery. (C) Immediately after surgery. (D) Six months after surgery in the same patient. 
Table 1.Summary of patients in the two groups according to the application of preoperative NPWT Table 2.Associations between complications and various factors References1. Kairinos N, Voogd AM, Botha PH, et al. Negative-pressure wound therapy II: negative-pressure wound therapy and increased perfusion. Just an illusion? Plast Reconstr Surg 2009;123:601-12.
2. Ichioka S, Watanabe H, Sekiya N, et al. A technique to visualize wound bed microcirculation and the acute effect of negative pressure. Wound Repair Regen 2008;16:460-5.
3. Moues CM, Vos MC, van den Bemd GJ, et al. Bacterial load in relation to vacuum-assisted closure wound therapy: a prospective randomized trial. Wound Repair Regen 2004;12:11-7.
4. Labler L, Mica L, Harter L, et al. Influence of V.A.C.-therapy on cytokines and growth factors in traumatic wounds. Zentralbl Chir 2006;131 Suppl 1:S62-7.
5. Barbera F, Lorenzetti F, Marsili R, et al. The impact of preoperative negative-pressure wound therapy on pectoralis major muscle flap reconstruction for deep sternal wound infections. Ann Plast Surg 2019;83:195-200.
6. Haesler E. Prevention and treatment of pressure ulcers/injuries: quick reference guide. European Pressure Ulcer Advisory Panel, National Pressure Injury Advisory Panel and Pan Pacific Pressure Injury Alliance (EPUAP/NPIAP/PPPIA); 2019.
7. Fleischmann W, Strecker W, Bombelli M, et al. Vacuum sealing as treatment of soft tissue damage in open fractures. Unfallchirurg 1993;96:488-92.
8. Morykwas MJ, Argenta LC, Shelton-Brown EI, et al. Vacuum-assisted closure: a new method for wound control and treatment: animal studies and basic foundation. Ann Plast Surg 1997;38:553-62.
9. Argenta LC, Morykwas MJ. Vacuum-assisted closure: a new method for wound control and treatment: clinical experience. Ann Plast Surg 1997;38:563-76.
10. Capobianco CM, Zgonis T. An overview of negative pressure wound therapy for the lower extremity. Clin Podiatr Med Surg 2009;26:619-31.
11. Venturi ML, Attinger CE, Mesbahi AN, et al. Mechanisms and clinical applications of the vacuum-assisted closure (VAC) device: a review. Am J Clin Dermatol 2005;6:185-94.
12. White RA, Miki RA, Kazmier P, et al. Vacuum-assisted closure complicated by erosion and hemorrhage of the anterior tibial artery. J Orthop Trauma 2005;19:56-9.
|
|