 |
 |

|
| Aims and Scope |
| About the Journal |
| About the Society (KWMS) |
| Editorial Board |
| Open Access |
| Principles of Transparency and Best Practice |
| Editorial Office |
| Subscriptions |


|
| Aims and Scope |
| About the Journal |
| About the Society (KWMS) |
| Editorial Board |
| Open Access |
| Principles of Transparency and Best Practice |
| Editorial Office |
| Subscriptions |

AbstractBackgroundStromal vascular fraction (SVF), which plays a substantial role in wound healing, has been discussed in many recent studies concerning its positive effect on scar formation. Our study explored the effects of SVF on scar formation from partially-removed transverse rectus abdominis muscle (TRAM) flaps after inset.
MethodsFrom December 2017 to May 2020, we enrolled 11 patients undergoing breast reconstruction performed by a single surgeon using the free TRAM flap. As a split-body, placebo-controlled study, SVF was subcutaneously injected into one side of the abdomen, and normal saline was injected into the other side of each patient. Scarring was evaluated using the Vancouver Scar Scale (VSS) and Patient Scar Assessment Questionnaire (PSAQ) at 1, 6, and 12 months after surgery, and histology was evaluated with immunofluorescence analysis at 6 months after surgery.
ResultsNo statistically significant differences were noted in the total scores or subcategory score of the VSS and PSAQ between the test and control groups. Some patients showed more positive staining for alpha smooth muscle actin, collagen type I, and type III in the test group than in the control group. However, quantification of positively stained areas showed no statistically significant difference.
ConclusionIntraoperative SVF injection had no demonstrable clinical effect on scar quality. Histology with immunofluorescence analysis also failed to demonstrate any significant effect of SVF on scars at the microscopic level. Despite previous studies indicating the positive effects of SVF on scar quality, this pilot study questions its true effectiveness.
IntroductionPrevention of hypertrophic scars and improvement in scar quality are of general concern among patients undergoing any type of surgical procedure. While the exact mechanism of hypertrophic scarring or keloid formation remains unknown, various topical scar management products are used in the postoperative period, targeting the wound healing phases. However, current clinical data from randomized control trials investigating the efficacy of topical scar management products is limited [1]. Therefore, many studies have sought methods to prevent hypertrophic scar formation in the intraoperative phase.
Abdominal flaps have unique advantages in breast reconstruction due to their autologous use, with an additional benefit of abdominoplasty. However, even with the best effort, donor site morbidities, such as long horizontal scars and dog ears, are inevitable. Niddam et al. [2] revealed that 34% of patients undergoing breast reconstruction with deep inferior epigastric perforator flaps were unsatisfied with the esthetic outcomes and 12% of them showed particular dissatisfaction with horizontal scars.
Stromal vascular fraction (SVF) is a cellular extract comprising a heterogeneous cell population. Its regenerative value arises from the potential differential and paracrine effects of adipose-derived stem cells (ADSCs) and various growth factors such as vascular endothelial growth factor. In addition to its well-known value in regenerative medicine, SVF is rapidly gaining recognition in esthetic fields. Several studies have demonstrated the positive effects of intralesional SVF injections on scar formation [3-7]. These studies have confirmed a gradual improvement in the Patient and Observer Scar Assessment Scale scores, with the most significant improvement noted in patient satisfaction, a gradual improvement noted in skin quality, and improvements noted in hyperpigmentation and pliability.
The present study evaluated the effect of intralesional SVF injections on scar formation in patients undergoing breast reconstruction with free transverse rectus abdominis muscle (TRAM) flaps. SVF can be easily harvested from partially removed flaps after inset without any additional procedure-related morbidities. Intraoperatively prepared automated closed systems using enzymatic methods can readily isolate SVF at the time of donor site closure without any delay in surgical time.
MethodsStudy design and settingThe study was a randomized, prospective, double-blinded, split-body, placebo-controlled study conducted by a single surgeon (USJ) between December 2017 and May 2020. Patients were randomly classified into two groups; in group A, SVF was injected at the right side of the abdomen while normal saline was injected at the left side and in group B, the injection side was reversed. Block randomization was performed using R script version 3.3.3 (R Foundation for Statistical Computing, Vienna, Austria) and the data was revealed to the operator who injected SVF. Information regarding the site of SVF injection was maintained blind to both the patient and the observer who assessed scar quality. Written informed consent was obtained before the procedure from all patients undergoing the treatment. The study was conducted in accordance with the Declaration of Helsinki, and the entire investigational protocol was approved by the Institutional Review Board of Seoul National University Hospital (IRB No. 1709-006-881).
Patient selection and criteriaThis study included female patients aged 20 to 70 years undergoing breast reconstruction with the free TRAM flap procedure, all of whom had provided written informed consent. Exclusion criteria were systemic metastases of breast cancer; history of immunosuppressants, cytotoxic agents, corticosteroids, and anticoagulants; metabolic disease; mental disorders; or little abdominal fat considered insufficient for isolation of SVF from the partially removed flap portion after inset.
Preparation and administration of SVFAmong the various isolation methods, our main objectives were as follows: (1) a short harvesting time that would not delay any surgical procedures and (2) a high cell yield and concentration for optimal outcomes. As the time between flap harvesting and donor site closure does not exceed 2 hours, the entire SVF isolation procedure should not exceed this time. We used the Cellunit SVF Isolation System (CG Bio Inc., Seongnam, Korea) for enzymatic SVF isolation with a closed automated system. Part of the harvested flap is usually removed after inset, especially from zone IV which is most prone to perfusion-related complications. Abdominal adipose tissue from the partially removed flap after inset was aspirated using a 3-mm inner diameter cannula attached to a 50-mL syringe. The aspirated fat tissue was collected in a chamber and was enzymatically digested with 0.1% collagenase (SERVA Electrophoresis GmbH, Heidelberg, Germany). The digested solution was washed with saline to remove any remnant collagenase and was centrifuged to refine SVF cells in the same chamber. In a previous study using a murine model, toxicity from remnant collagenase activity and tumorigenicity was insignificant after one washing step [8]. Therefore, the use of collagenase could be considered safe for isolation of SVF. SVF was collected within 45 minutes using the automated system. Additional centrifugation was performed to concentrate the suspension to 5 mL. From two out of eleven donors, we recorded the number of nucleated SVF cells using an automated cell counter (LunaFLTM; Logos Biosystems, Anyang, Korea). The final concentration was 32 and 8.2×105 cells/mL and the cell viability of isolated SVF cells was 82.5% and 87.9% each. While the cell suspension was being prepared, the donor site wound was closed layer by layer beginning from Scarpa’s fascia to dermal layer, using Monosyn 2-0 and 3-0. After some skin sutures for precise epidermal contact, 0.5 mL of SVF was injected into the intradermal layer of both sides of the suture at 1-cm intervals in one side of the abdomen, amounting to a total of 10 mL of SVF. The same procedure was performed with normal saline on the other side of the abdomen (Fig. 1). Then, self-adhering mesh with dermal bond (Dermabond Prineo; Ethicon US LLC, Cincinnati, OH, USA) was used for final skin closure. Following our institution’s protocol for abdominoplasty wound management, the patient’s hip joint was kept at a flexed position until the patient could feel no tension at her abdomen. Prineo and stitch removal was performed at postoperative day 14, with no additional application of scar-related products.
Scar assessmentIn all the patients, demographic data related to scar quality were collected preoperatively, and the scars were scored immediately after surgery and at 1, 6, and 12 months after surgery. Biopsy was performed using a 4 mm punch biopsy at postoperative 6 months. Scar assessments were performed by a single observer who did not participate in the operation. For objective scar assessment, the Vancouver Scar Scale (VSS) was used, and the data were obtained by a single examiner through direct inspection at the outpatient clinic. For subjective assessment of satisfaction, the Patient Scar Assessment Questionnaire (PSAQ) was used. The assessment was explained to the patients and performed by the same examiner.
Hematoxylin and eosin (H&E), Masson’s trichrome, and immunofluorescence stainingAll the 22 obtained samples from the test (SVF) and control groups were fixed in 10% neutral buffered formalin, prepared as paraffin blocks, and cut into 3-μm-thick sections. The sectioned slides were deparaffinized and hydrated using ethyl alcohol at 70% concentration. H&E and Masson’s trichrome staining was performed after the slides were deparaffinized. For immunofluorescence staining, antigen retrieval was conducted at 20°C to 25°C for 15 minutes with slides on which the hydration process was completed using Proteinase K solution (1:10 dilution). Then, the slides were washed twice with distilled water for 1 minute and once with phosphate-buffered saline (PBS) for 1 minute. Endogenous peroxidase activity was blocked by incubation in 3% H2O2 for 10 minutes, and the slides were washed with PBS for another 3 minutes. Serum blocking was performed by incubating the sections with 10% normal serum albumin for 30 minutes to block nonspecific immunoglobulin binding. Then, we incubated the sections with primary antibodies at appropriate dilutions in PBS for 1 hour at room temperature.
The primary antibodies used in this study were directed against alpha smooth muscle actin (αSMA, 1:200, ab7817; Abcam, Cambridge, UK) to stain smooth muscle cells, vimentin (1:250, ab92547; Abcam) to stain myofibroblasts, and collagen type I (1:100, ab34710; Abcam) and collagen type III (1:200, ab7778; Abcam) to stain collagen fibers. The slides were washed thrice in PBS with 0.5% Tween20 for 5 minutes each. The sections were then incubated for 1 hour at room temperature with fluorochrome-conjugated antibodies diluted in PBS (1:200 dilution). The sections were stored in a dark humid chamber. The secondary antibodies used in this study were Alexa Fluor 594 antibody (ab150116; Abcam) for αSMA and vimentin to develop a red color and Alexa Fluor 488 antibody (ab150077; Abcam) for collagen type I and III to develop green color. The slides were washed again thrice in PBS with 0.5% Tween20 for 5 minutes each. Counterstaining was performed using 4´,6-diamidino-2-phenylindole.
Image quantificationCaptured images of immunofluorescence staining were analyzed using ImageJ (National Institutes of Health, Bethesda, MD, USA) for quantification of positive staining areas. First, the outlines of the dermis areas were marked manually, and the areas were analyzed using ImageJ. The areas of empty spaces such as vessel lumens and vacuoles were subtracted. Then, the images were converted to grayscale (click Image, then click Type, then click 8-bit) and adjusted to the auto-threshold (click Image, then click Adjust, then click Auto threshold, then click Method: Default) to mark positive staining areas (Fig. 2). The percentage of positive staining areas was then calculated.
Statistical analysisThe result of our study is always a paired sample, and we applied the Shapiro-Wilk test to check if the data followed the normal distribution (P>0.05). When the data followed the normal distribution, paired t-test was applied and if not, Wilcoxon’s signed rank test was applied. Results of scar assessment methods (i.e., VSS and PSAQ) at each point for the test and control groups and immunofluorescence quantification data were analyzed following the method described above. The time effect of each variable was analyzed using repeated measures analysis of variance (RM-ANOVA). The t-tests were performed using the SAS system version 9.4 (SAS Institute, Cary, NC, USA).
Two patients failed to make their last visit at 12 months, resulting in two missing values of scar quality assessment: VSS and PSAQ. We replaced these subjects’ previously observed value in accordance with the last observation carried forward method.
ResultsBetween December 2017 and May 2020, 11 female patients with an average age of 58.8±3.2 years were included in this study. All the patients underwent breast reconstruction with a free TRAM flap performed by a single surgeon, and seven patients (63.6%) underwent concomitant mastectomy. Seven patients (63.6%) had a medical or surgical history and nine patients (81.8%) had concomitant medication. None of the patients developed hypertrophic scars or keloids 12 months after surgery. The patient demographics are summarized in Table 1.
Scar scoreNone of the total scores or any subcategory score of the VSS showed statistical significance (Table 2). In the RM-ANOVA results, time effects for the following subcategories showed a decrease with statistical significance: vascularity, height, and total score (Table 3). Further, no statistical significance was noted between the total scores or any subcategory score in the PSAQ (Table 4). The RM-ANOVA showed statistical significance for time effect with regard to the appearance/symptom subcategory (Table 5).
H&E, Masson’s trichrome, and immunofluorescence stainingH&E staining showed nonspecific dermal fibrosis, a typical appearance of scar tissue. Masson’s trichrome staining showed collagen deposits in the fibrotic areas (Fig. 3). Immunofluorescent images showed diffuse staining of αSMA, vimentin, and collagen types I and III in the test (SVF) and control groups (Fig. 4). Comparison of slides for the same patient indicated that some patients displayed different staining areas (Fig. 5). Quantification of the positively stained areas using ImageJ revealed that in the test group, eight patients showed a larger positively stained area for αSMA (72.7%), and nine patients showed a larger positively stained area for collagen types I and III (81.8%). Conversely, in the control group, eight patients (72.7%) showed a larger positively stained area for vimentin (Table 6). However, none of the results for any of the markers showed statistical significance (Table 7, Fig. 6).
DiscussionAt present, cell-based therapies are trending in the field of reconstructive surgery. Surgeons can easily access lipoaspirate products through liposuction, and ADSCs and SVF are gaining rapid momentum in reconstructive surgery and esthetic fields. In clinical application, SVF has an advantage over ADSCs in that it does not require a culture process and, therefore, does not require U.S. Food and Drug Administration approval, thereby making it readily available. Therefore, expanding clinical indication of SVF will allow numerous patients to benefit.
Collagenase-based digestion is the most effective method to isolate SVF. Markarian et al. [9] reported about 350,000 nucleated cells/cc using the enzymatic method, while the non-enzymatic method using centrifugation of lipoaspirate generated only about 10,000 nucleated cells/mL. Although the use of collagenase is still dubious in several countries, there is a recent study supporting the safety of collagenase in terms of in vivo toxicity and tumorigenicity [8]. With increasing demand for optimizing SVF harvest, an automated closed-system SVF was developed to reduce the human element, increase sterility and limit loss of cell viability from processing. A summary comparing manual versus automated closed systems using the enzymatic method showed no difference in cell yield and viability [10].
Previous studies regarding intralesional SVF injection did not suggest effective dosages with exact cell counts for scar quality improvement; the study performed by Lee et al. [11] used 2-mL SVF pellets obtained by manual SVF isolation using the enzymatic method without any information of cell count and cell viability. A pilot study by Moon et al. [12] describes the beneficial effects of intralesional SVF injection on microcirculation in ischemic diabetic feet with the same SVF isolation method used in this study. The final concentration of SVF cell suspension was 9.6±4.2×105 cells/mL (range, 4.8×105 to 16.8×105 cells/mL) and the injected dose of the SVF cell suspension was 0.3 mL per site. In our study, Cellunit generated similar concentration of SVF cell suspension. Also, 0.5 mL of SVF cell suspension was injected per site, which we believed to be sufficient for generating similarly beneficial effects of SVF injection.
While the exact mechanism of SVF on scar quality is unknown, there are some reports indicating that stem cells could improve hypertrophic scars. Lee et al. [11] demonstrated the clinical efficacy of SVF in surgical management of contracted and depressed scars and explained their results by applying these properties in different stages of wound healing. All the patients in the test group of Lee et al.’s study showed improvement in parameters of various scar assessment scales, especially in terms of height and pliability, but no significant changes were noted in vascularity [11]. Another study investigating the paracrine effects of bone marrow-derived mesenchymal stem cells (BMSCs) reports that BMSCs reduce the proliferative and profibrotic phenotype associated with hypertrophic scar fibroblasts and keloid fibroblasts and inhibit extracellular matrix (ECM) synthesis through a paracrine signaling mechanism [13].
While most recent studies focus on the beneficial effects of stem cells on wound healing and preventing hypertrophic scar formation, another study warned about the use of stem cell therapy in wound healing, especially in patients susceptible to keloids or hypertrophic scars [14]. Keloid fibroblasts were reported to be chemotactic to human mesenchymal stem cells in vitro and their co-culture showed keloid fibroblast phenotype in human MSC [15]. This differentiation capacity could exaggerate the proliferation of myofibroblasts and upregulate the production of ECM proteins, leading to scar hypertrophy.
This is the first study designed to analyze the effect of SVF on scar formation with scar assessment scales and histologic analysis; other studies only analyzed the clinical outcomes of SVF application [3-7,11] and studies with histologic analysis were based on cultured stem cell therapies [13,16]. A previous study hypothesized that in the remodeling phase, excessive synthesis of collagen is suppressed by chemokines such as the transforming growth factor β-3 and matrix metalloproteinases [17]. Shorter inflammation phases and faster tensile strength restoration could result in better scar quality. However, another study has suggested that ADSCs enhance the secretion of collagen type I in human dermal fibroblasts by regulating the mRNA levels of ECM proteins, including the upregulation of collagen type I [15]. In our study, immunofluorescence imaging for αSMA, vimentin, and collagen types I and III revealed some differences in the positively stained areas between the test and control groups. Immunofluorescence positive stained areas were quantified using ImageJ, and more patients showed positively stained areas for αSMA and collagen types I and III in the test (SVF) group than in the control group. In contrast, more patients showed positively stained areas for vimentin in the control group than in the test group. However, none of the results showed a statistically significant difference, suggesting that intralesional SVF injection had no significant effect on scars at the microscopic level; neither excessive synthesis nor downregulation of collagen synthesis were explained.
Our study failed to demonstrate any effect of SVF on scar quality, both clinically and histologically. As a pilot study, it had some limitations: (1) a relatively small number of participants were included; (2) as histological analysis was performed only once in the remodeling phase, additional histological analysis in the inflammatory and proliferative phases could help better explain the physiology of SVF and its effect on scar formation; (3) SVF was injected only once, which may not be an effective dose, despite the fact that it was highly concentrated through additional centrifugation; and (4) the scar was observed and assessed only by a single observer. Despite the limitations, our study has its strength in that it was an attempt to estimate the effect of SVF with least scar-related variables; other studies which reported positive SVF values on various types of scars differed in their location, etiology and onset [3-7]. Our design allows comparison of the wounds as a paired sample, and all the patients underwent the same procedure by a single surgeon. For this reason, we strongly suggest that intralesional SVF injection could not improve scar quality as expected. However, the small number of patients limit this suggestion, which is why we report this result as a pilot study. We concluded that further study is required to reveal the true effect of SVF on scar formation.
Conflict of InterestThis work was supported by CG Bio Inc. (Seongnam, Korea) for the use of Cellunit SVF Isolation System and financial support. No other potential conflicts of interest relevant to this article were reported. Fig. 1.Intraoperative injection in test and control group after wound closure. Total amount of 10 mL of stromal vascular fraction (SVF) for the test group and saline for the control group was injected intraoperatively. Suture materials and methods were the same. 
Fig. 2.Immunofluorescence staining positive area calculation with ImageJ (×40). (A) Area of dermis was outlined manually, subtracting areas for empty lumens or vacuoles. (B) Ratio of staining positive area was calculated with Image J by adjusting the image to 8 bit and applying auto-threshold. 
Fig. 3.Diffuse dermal fibrosis and collagen deposits. (A) H&E staining (×40). (B) Masson’s Trichrome staining (×40). 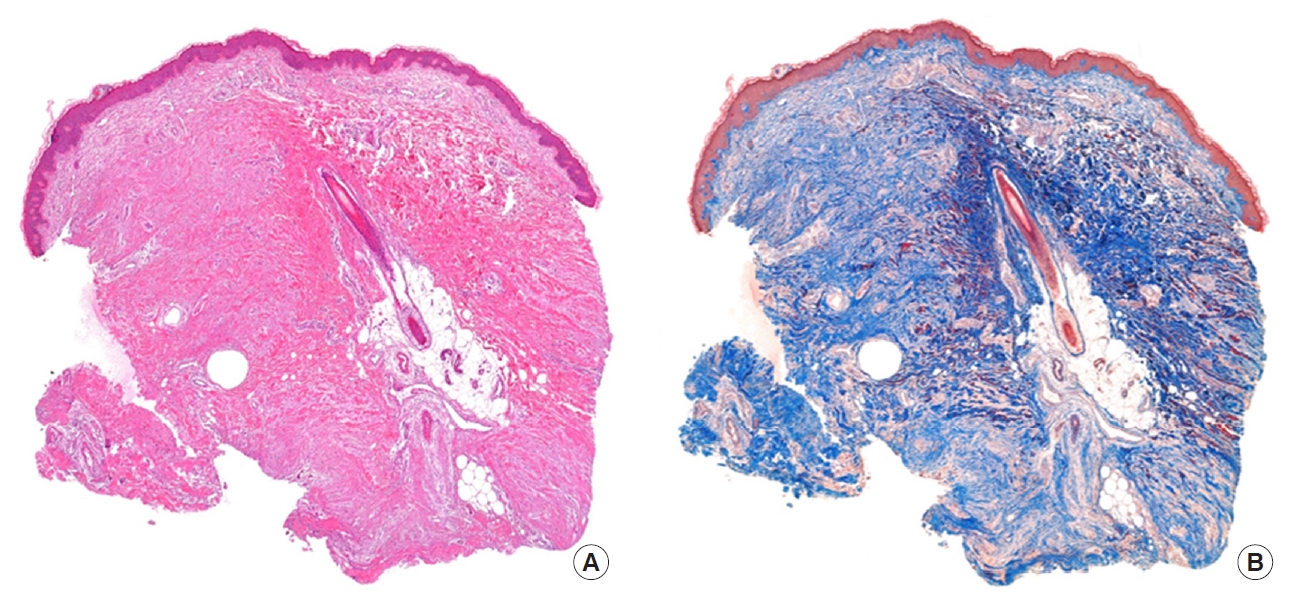
Fig. 4.Immunofluorescence images of four types of staining (×100). The positive staining areas are marked with yellow arrows. (A) Alpha smooth muscle actin. (B) Vimentin. (C) Collagen type I. (D) Collagen type III. 
Fig. 5.Positive staining areas for each marker. All slides were obtained from the same patient (×40). 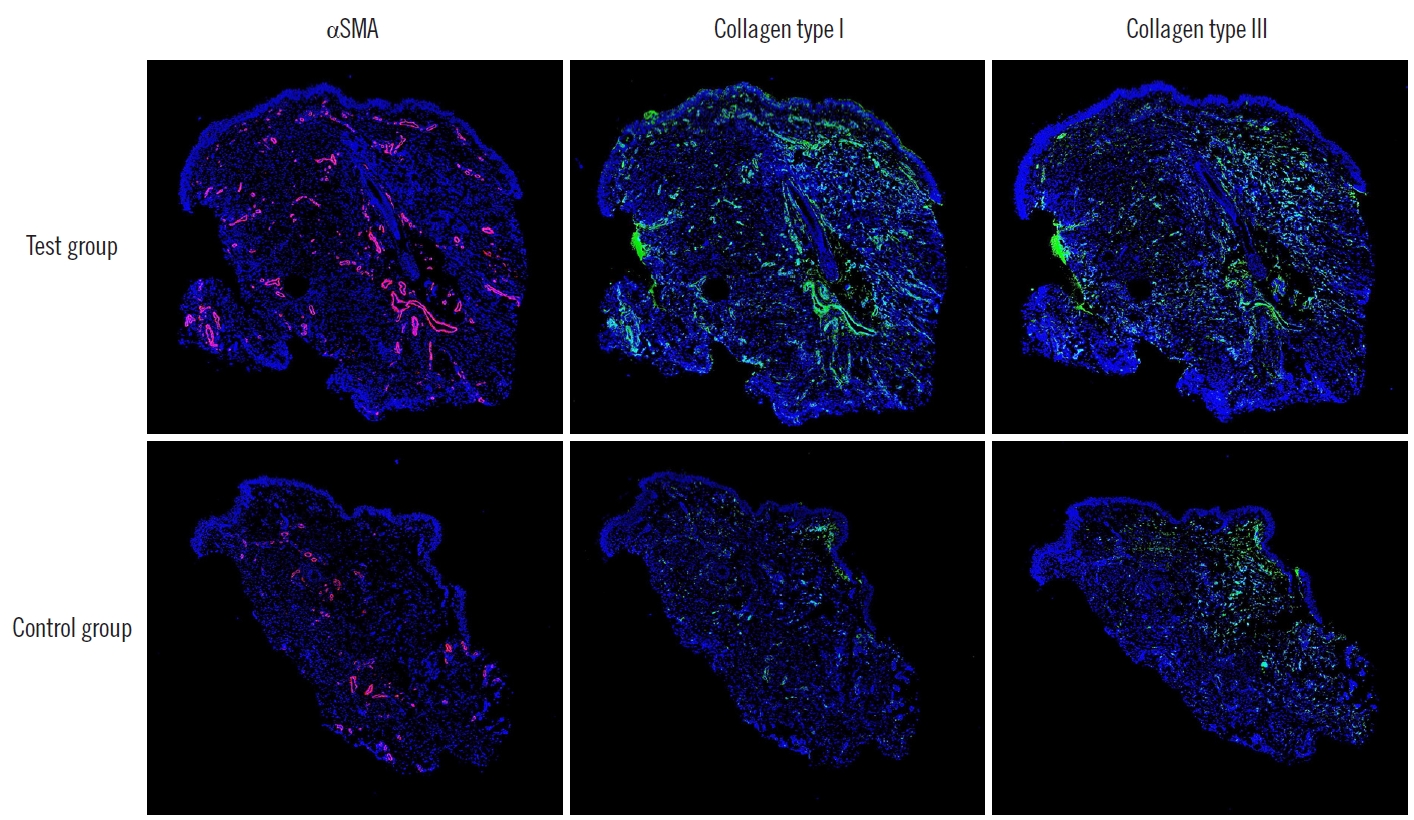
Fig. 6.Statistical analysis of quantification data. No result for any marker showed difference with statistical significance. αSMA, alpha smooth muscle actin. 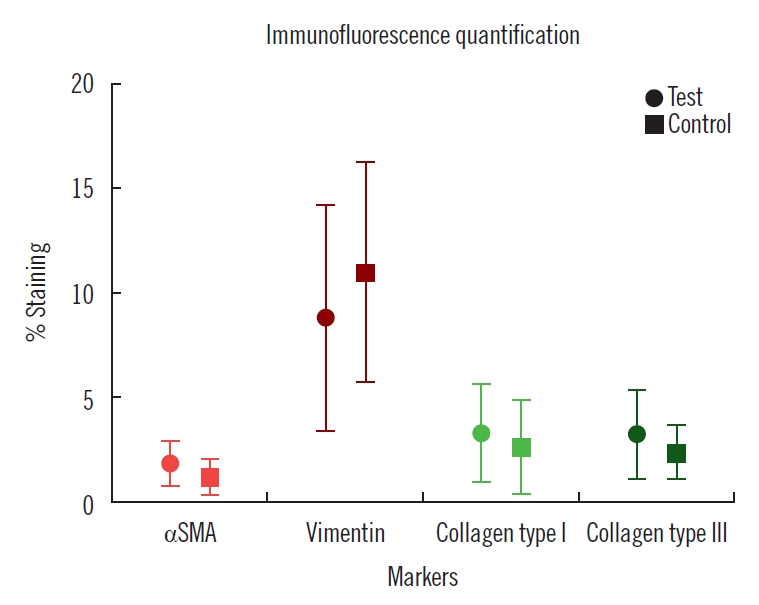
Table 1.Patient demographics Table 2.Vancouver Scar Scale (VSS) assessment with full analysis set
Table 3.Vancouver Scar Scale (VSS): repeated measures analysis of variance result Table 4.Patient Scar Assessment Questionnaire (PSAQ) assessment with full analysis set
Table 5.Patient Scar Assessment Questionnaire (PSAQ): repeated measures analysis of variance result Table 6.Quantification of immunofluorescence positive areas
References1. Tran B, Wu JJ, Ratner D, et al. Topical scar treatment products for wounds: a systematic review. Dermatol Surg 2020;46:1564-71.
2. Niddam J, Bosc R, Lange F, et al. DIEP flap for breast reconstruction: retrospective evaluation of patient satisfaction on abdominal results. J Plast Reconstr Aesthet Surg 2014;67:789-96.
3. Bhooshan LS, Devi MG, Aniraj R, et al. Autologous emulsified fat injection for rejuvenation of scars: a prospective observational study. Indian J Plast Surg 2018;51:77-83.
4. Gu Z, Li Y, Li H. Use of condensed nanofat combined with fat grafts to treat atrophic scars. JAMA Facial Plast Surg 2018;20:128-35.
5. Jan SN, Bashir MM, Khan FA, et al. Unfiltered nanofat injections rejuvenate postburn scars of face. Ann Plast Surg 2019;82:28-33.
6. Tenna S, Cogliandro A, Barone M, et al. Comparative study using autologous fat grafts plus platelet-rich plasma with or without fractional CO2 laser resurfacing in treatment of acne scars: analysis of outcomes and satisfaction with FACE-Q. Aesthetic Plast Surg 2017;41:661-6.
7. Gentile P, Scioli MG, Bielli A, et al. Comparing different nanofat procedures on scars: role of the stromal vascular fraction and its clinical implications. Regen Med 2017;12:939-52.
8. Chang H, Do BR, Che JH, et al. Safety of adipose-derived stem cells and collagenase in fat tissue preparation. Aesthetic Plast Surg 2013;37:802-8.
9. Markarian CF, Frey GZ, Silveira MD, et al. Isolation of adipose-derived stem cells: a comparison among different methods. Biotechnol Lett 2014;36:693-702.
10. Aronowitz JA, Lockhart RA, Hakakian CS. Mechanical versus enzymatic isolation of stromal vascular fraction cells from adipose tissue. Springerplus 2015;4:713.
11. Lee JW, Park SH, Lee SJ, et al. Clinical impact of highly condensed stromal vascular fraction injection in surgical management of depressed and contracted scars. Aesthetic Plast Surg 2018;42:1689-98.
12. Moon KC, Chung HY, Han SK, et al. Possibility of injecting adipose-derived stromal vascular fraction cells to accelerate microcirculation in ischemic diabetic feet: a pilot study. Int J Stem Cells 2019;12:107-13.
13. Fang F, Huang RL, Zheng Y, et al. Bone marrow derived mesenchymal stem cells inhibit the proliferative and profibrotic phenotype of hypertrophic scar fibroblasts and keloid fibroblasts through paracrine signaling. J Dermatol Sci 2016;83:95-105.
14. Lau K, Paus R, Tiede S, et al. Exploring the role of stem cells in cutaneous wound healing. Exp Dermatol 2009;18:921-33.
15. Nangole FW, Agak GW. Keloid pathophysiology: fibroblast or inflammatory disorders? JPRAS Open 2019;22:44-54.
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||