Introduction
Cosmetic and functional restoration of tissue defects is a major challenge faced by re-constructive surgeons. Microsurgical free tissue transfer has become an increasingly common technique for various soft tissue reconstructions, resulting in satisfying out-comes with reported success rates of 96% to 99%, while exploration for vascular compromise is required in between 2% to 5% [1]. Over the past few decades, the success rate of flaps has increased, and consequently the number of cases requiring revision due to vascular compromise has decreased [1].
However, in such frustrating situations where free flaps do fail, since a large amount of tissue has already been harvested in cases with a large defect, further consideration of the donor site through tissue preservation or recycling is warranted in revision surgery. If skin from the failing flap can be recycled for subsequent wound closure, the need for additional donor sites can be minimized, with cosmetic and pain reduction advantages. However, the recyclability of salvaged skin is mainly dependent on warm ischemia time [2].
In this case study, split-thickness skin harvested from a failed flap with an ischemic time of more than 48 hours was successfully recycled, indicating the feasibility of this technique. Written informed consent was obtained from the patient for the publication and use of photographs and videography.
Idea
Patient and surgical techniques
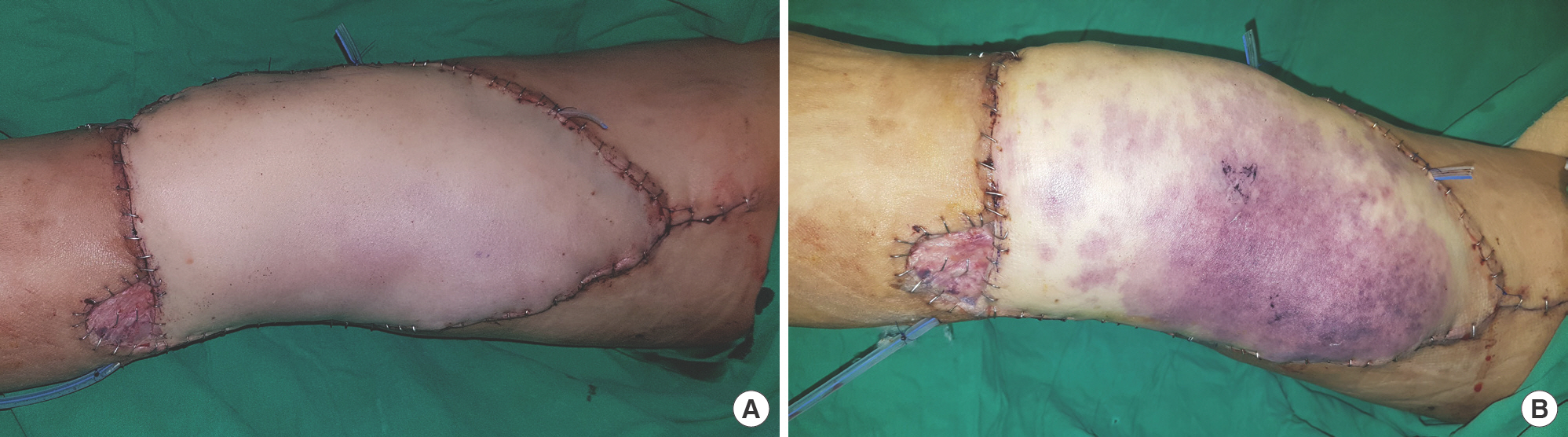
A 59-year-old man without any underlying disease except Guillain-Barré syndrome, which was completely cured, pre-sented to the outpatient clinic with a scar contracture deformity on his right medial thigh. After the 18×30-cm-sized con-tracted scar tissue was excised, the remaining defect was re-constructed with an 18×32-cm-sized thoracodorsal artery perforator free flap harvested from the right flank (Fig. 1A). Unfortunately, flap compromise was found on the second postoperative day; an attempt to salvage the flap through emergent surgical exploration ultimately failed due to extensive intravascular thrombus (Fig. 1B). Another thoracodorsal artery perforator free flap was harvested from the left flank for revision. The second donor site was partially primarily closed after undermining; however, as a large amount of tissue had been used, a split-thickness skin graft (STSG) harvested from the failed previous flap was planned to cover the remaining defect.
Fig. 1.
Failure of the first free flap. (A) Immediate postoperative photo after the first flap surgery. No flap compromise was found until the day after. (B) Two days after the first flap surgery. Flap compromise was found. Venous congestion was suspected based on the purplish color change in most of the flap area and increased tension.
A 15×30-cm-sized STSG was harvested with a thickness of 12/1,000 inches with a dermatome (Zimmer Air Dermatome; Zimmer Biomet, Zug, Switzerland) from the failed flap. Seven hand-held retractors, consisting of single or double-pronged skin hooks, were utilized in all directions to provide appropriate and uniform tension for harvesting the STSG (Supplementary Video 1). The harvested skin was meshed by a 1:1.5 ratio using a skin graft mesher (MeshGraft II Tissue Expansion Sys-tem; Zimmer Biomet) and grafted with acellular dermal matrix (MatriDerm; MedSkin Solutions Dr. Suwelack AG, Biller-beck, Germany) into the remaining donor defect. A compres-sive dressing with negative wound pressure therapy (NPWT) (INFOV.A.C.; Kinetic Concepts, Inc., San Antonio, TX, USA) set at 125 mmHg was applied.
Postoperative management
NPWT for grafted skin was maintained for 5 days and then removed (Fig. 2A), followed by daily dressings with careful ap-plication of medicated paraffin gauze (Bactigras; Smith & Nephew, New South Wales, Australia) and ointment. The con-dition of the grafted skin and the presence of any problems such as infection, hematoma, or seroma were evaluated when NPWT was removed and in every subsequent dressing.
Fig. 2.
Photos after split-thickness skin graft harvested from the failed flap. (A) Postoperative day 5. After removal of negative pressure wound therapy, the graft was well attached to the flap donor site, but only minimal additional epithelization was observed. (B) Postoperative day 14. Several unhealed points are found at the engrafted thoracodorsal artery perforator flap donor site. (C) Postoperative day 21. The engrafted skin was eventually well taken without gross problems.
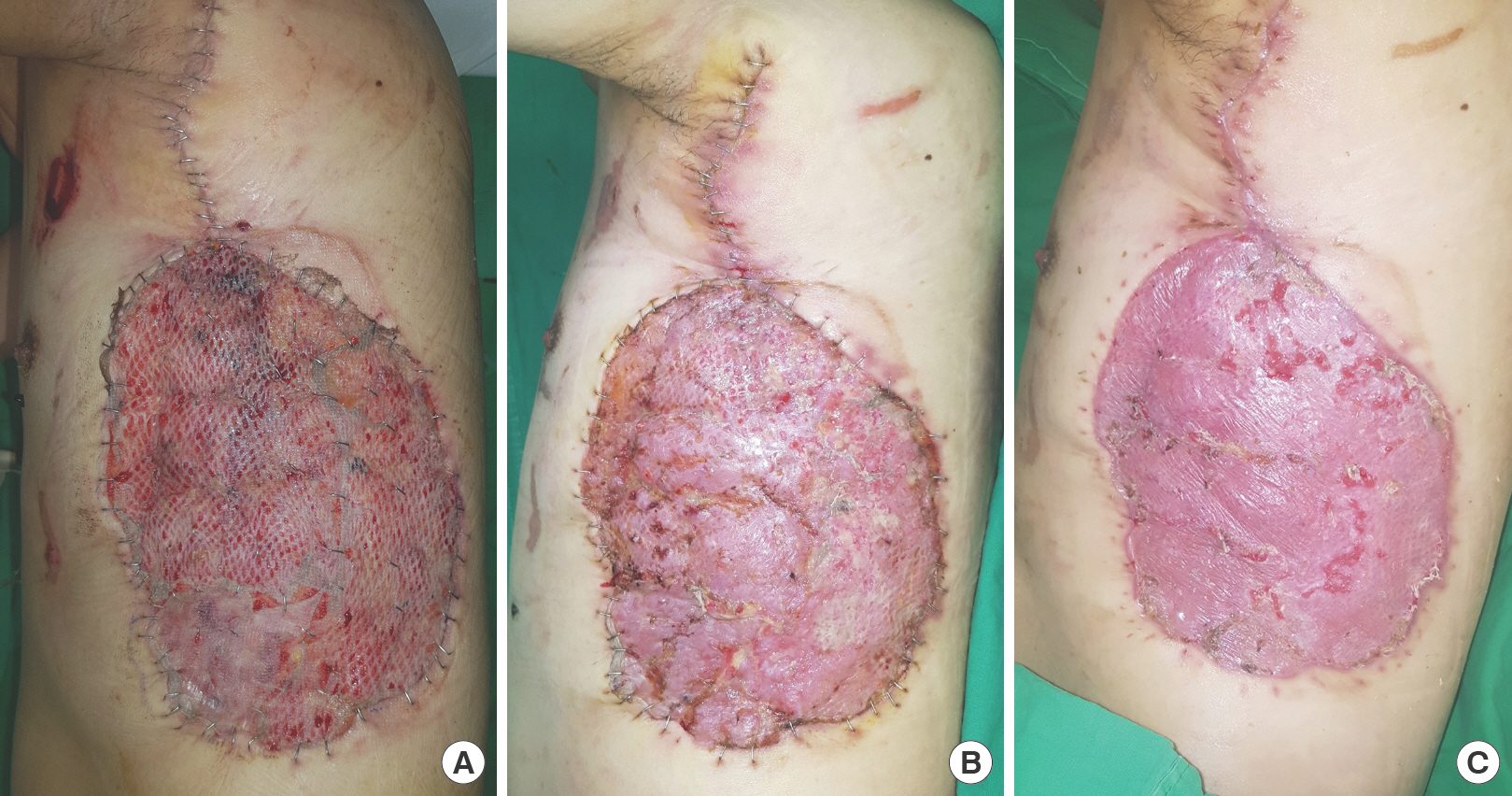
Two weeks after surgery, the overall skin graft site was pro-gressively healing, but small non-taken areas were observed in several regions (Fig. 2B). After an additional week, the patient was discharged on crutches with almost complete healing of the skin graft site (Fig. 2C). Although the estimated ischemic time of the first flap was more than 48 hours, STSG with skin harvested from the failed free flap was successful.
Discussion
Several surgeons have attempted to recycle skin from flaps. In 2007, Lowrie et al. [3] described a case of harvesting STSG from the skin of a pedicled myocutaneous latissimus dorsi flap while utilizing the flap simultaneously. The “bowtie scar” method was also introduced by Ha et al. [4] as a slightly modi-fied method for harvesting a STSG to include the overlying skin while elevating the gracilis free flap. These methods focused on minimizing morbidity, including donor site pain and scarring.
More recent studies focus on “tissue recycling” from both healthy and failed previously performed flaps. In 2011, Tan et al. [5] introduced tissue recycling from a previous flap for cor-recting independent flexion contracture, while debulking the flap simultaneously. Chadwick and Kosutic [6] created a graft-able wound bed using a failed necrotized flap after delayed de-bridement, and then completed reconstruction using a skin graft. Sadigh et al. [7] introduced a “green” approach, recycling previously used flaps in 60 cases and classifying recycled flaps according to whether they were random pattern flaps, pedicled perforator flaps, flaps involving pedicle skeletonization, or free perforator flaps. Hallock [8] also described a case with a recycled keystone flap, which was classified as a type II recycled flap in the schema of the “green” approach.
Similar to our case, Chen et al. [9] proposed the idea of harvesting skin grafts from failed flaps, although they focused on minimizing donor sites. They performed 14 cases of full-thickness skin graft (FTSG) harvesting from failed flaps and suggested that FTSG has advantages over STSG, but in two cases, an additional STSG was needed because the FTSG was too small. Since the patient in our report had a rather large defect at the flap donor site, an STSG was more suitable for cov-ering the defect.
Chen et al. [9] also tried to compare the viability of FTSGs by estimating the ischemic time of flaps. Although the grafts had an average warm ischemic time of 13.7 hours, which is longer than previously reported warm ischemic tolerance time of 6 to 8 hours for skin, all FTSGs were successfully engrafted. As STSGs are relatively less affected by ischemic time than FTSGs, and also because the graft was applied to a flap donor site with a healthy wound bed in our case, a high probability of success was anticipated even though the estimated ischemic time was longer than 48 hours [3].
On the other hand, the viability of skin is preserved for longer in relation to cold ischemia time. Since the 1960s, studies have been conducted on preserving skin at low temperatures, and it has been reported that cryopreserving maintains enough viability to allow grafting for more than 5 years [10,11].
An important part of our technique was harvesting split-thickness skin from a mobile and unstable failed flap after surgical exploration. The first step was to thin and flatten the flap by excising subcutaneous tissue, thus making it less resilient and bouncy. In the next step, multiple single or double-pronged skin hooks were used as hand-held retractors to cre-ate appropriate and uniform tension, enabling skin harvesting using a dermatome from the failed flap. The required number of retractors can vary according to the dimension and shape of the flap; it seems that at least six are necessary to sufficiently flatten the flap in all directions.
Previous studies on Matriderm (MedSkin Solutions Dr. Suwelack AG), the acellular dermal matrix used in this study, have reported that it has the advantages of restoring skin qual-ity and showing minimal complications, and a significant reduction in healing time with faster re-epithelialization [12,13]. In the case of meshed STSG, there is also a report that the acellular dermal matrix mitigates the fishnet appearance of the grafted site [14]. We have previously experienced successful engrafting with acellular dermal matrix by harvesting small STSGs less than 5×5 cm from failed flaps with a relatively short warm ischemia time of approximately 12 to 24 hours. On these grounds, STSG was also performed using acellular dermal matrix in this case.
However, the STSG in this study required a treatment peri-od of more than 3 weeks, which was longer than usual. This is presumed to be due to the decreased viability and metabolic activity of harvested skin from the extended warm ischemia time. To compensate for this, applying NPWT for longer than the customary 5 days is proposed for additional epithelization and stability of STSG [15].
It would be ideal if flap compromise could be detected quickly, explored, and resolved, but in cases of flap failure, recycling the skin portion from the failed flap has distinct advantages. Since this study showed the possibility of harvesting STSG from a failed flap with a long warm ischemia time, we encourage that surgeons employ this method to minimize tissue waste and maximize resource utilization.















