Effect of Retinoic Acid on Proliferation of Human Umbilical Vein Endothelial Cells
Article information
Abstract
Background
Although many recent studies are focusing on the role of retinoids as a treatment in wound healing, the exact principles and effects of retinoids in wound healing remain unclear. The purpose of this study was to examine the in vitro effects of retinoids on vascular endothelial cells, which play an important role in angiogenesis for wound healing.
Methods
We investigated in vitro the proliferative activities of human umbilical vein endothelial cells (HUVECs) in various concentrations of all-trans-retinoic acids (ATRAs). We treated the cells with four different concentrations of the acids and assessed the HUVEC proliferation by counting the cell populations and directly measuring the absorbance at 450 nm. We also assessed the HUVEC proliferation activity after adding 100 μg/mL and 1,000 μg/mL of the wound-healing promoter polydeoxyribonucleotide (PDRN) with ATRA.
Results
The absorbance in all groups was the highest on day 2, followed by decreasing tendency. Every group with ATRA except 1 nM group showed significantly lower absorbance than the control group from day 2 through day 9, and cell proliferation decreased with increasing concentrations of the ATRA (P<0.05). The proliferation activity of HUVEC also decreased in the media treated with ATRA and PDRN (P<0.05).
Conclusion
The ATRA in concentrations from 10 nM to 1,000 nM has suppressed the proliferation of vascular endothelial cells in vitro. Because vascular endothelial cells play a key role in wound healing, retinoic acid may delay wound healing. Further studies are needed to clear its negative effect on wound healing.
Introduction
Retinoic acids are polyisoprenoid compounds that contain a cyclohexynyl ring and are structural and functional analogues of vitamin A. Retinoic acid can be produced in the body by two sequential oxidation steps. All-trans retinol (vitamin A) is oxidized to produce all-trans retinal (retinaldehyde), and retinaldehyde is oxidized to produce all-trans retinoic acid, which is the main biologically active form of vitamin A. Retinoids exert their effects on gene transcription by binding to intracellular receptors in the nucleus and influence cell division, differentiation, RNA, and protein synthesis [1]. Because insufficient vitamin A supply can result in immunodeficiency, anemia, and abnormal cell differentiation, researcher interest has increased in the areas of dermatology, cancer research, and embryonal development. As a result, retinoids are used in treating acute promyelocytic leukemia, and recent studies report their effects on cardiovascular disease [2,3]. In contrast, other studies report that treating endothelial cells with isotretinoin (13-cis-retinoic acid) has increased the risk of atherosclerosis and hyperlipidemia [4,5].
There is also controversy about the role of vitamin A in wound healing. Isotretinoin is conventionally used to treat acne vulgaris and psoriasis, and pretreatment with retinoid is widely used before dermabrasion, chemical peels, and laser resurfacing [6]. These treatments are based on the hypothesis that retinoids accelerate wound healing by promoting angiogenesis in the epidermis and dermis and affecting cells in the inflammatory phase [7,8]. In contrast, some studies report that retinoids do not affect wound healing, and others have found that they even delay wound healing [9–11].
Many recent studies about the role of retinoids in wound healing have focused on fibroblasts and keratinocytes. In this preliminary study, we focused on vascular endothelial cells, which also play an important role in wound healing and thus, the human umbilical vein endothelial cells (HUVECs) that maintain nearly all of the features of native vascular endothelial cells were used.
Methods
In vitro proliferative activity of HUVECs in various concentrations of all-trans-retinoic acid (ATRA)
We purchased HUVECs from the American Type Culture Collection (ATCC, Manassas, VA, USA) and treated with four different concentrations of ATRA (Sigma-Aldrich, St. Louis, MO, USA) from 1 to 1,000 nM (1, 10, 100, and 1,000 nM). We cultured a total of 2×105 cells per well in a 6-well plate in endothelial basal medium (EBM-2, Lonza, Basel, Switzerland) supplemented with 10% fetal bovine serum (FBS) at 37°C and 5% CO2 for 24 hours. After 24 hours (day 0), we added 100 μL of Cell Counting Kit-8 (CCK-8; Dojindo, Kumamoto, Japan) solution to each well and incubated for 1.5 hours in the incubator. We measured the absorbance at 450 nm using a microplate reader. After changing 10% FBS to 1% FBS and adding 1 mL of ATRA in four different concentrations per well, we cultured the viable cells for another 24 hours. We repeated this sequence every 24 hours in the control group and in all four experimental groups.
In vitro proliferative HUVEC activity in RA with a wound healing promoter
We treated HUVECs with the wound healing promoter polydeoxyribonucleotide (PDRN, Placentex Integro®, Mastelli Srl-Sanremo, Italy) in concentrations of 100 μg/mL and 1,000 μg/mL. Per well, 5×104 cells were cultured in EBM-2 and 0.5% FBS at 37°C and 5% CO2 for 24 hours. After 24 hours (day 0), we added 100 μL of the CCK-8 solution to each well and incubated it for 1.5 hours in the incubator. We measured the absorbance at 450 nm with a microplate reader. We added 2 mL of PDRN to the viable cells in the control group and 2 mL of PD-RN with 1,000 nM ATRA to the viable cells in the experimental group. Adding 100 μL of CCK-8 followed by 1.5 hours incubation and absorbance measurement and adding 2 mL of PDRN or PDRN with ATRA sequence were repeated every 24 hours.
Statistical analysis
We compared the average absorbance in the control and experimental groups by day after seven repeated experiments. We analyzed all data by Kruskal-Wallis test (post-hoc Mann-Whitney test) using SPSS version 21.0 for Windows (SPSS; Systat Software, IBM, Armonk, NY, USA). We selected a probability error of <0.05 as the criterion for statistical significance.
Results
In vitro HUVEC proliferative activity in various concentrations of ATRA
Starting from the same absorbance on day 0, the absorbance had increased until day 2 and began to decrease from day 3 in all five groups, which lasted until the end of the experiment (Fig. 1 and Table 1). Comparing the control group with each experimental group, there was statistically significant difference each day (P<0.001) except the 1 nM ATRA group. Comparing the four experimental groups, the absorbances decreased significantly as the ATRA concentrations increased (P<0.001). However, there was no statistically significant difference between 1 nM and 10 nM RA groups.
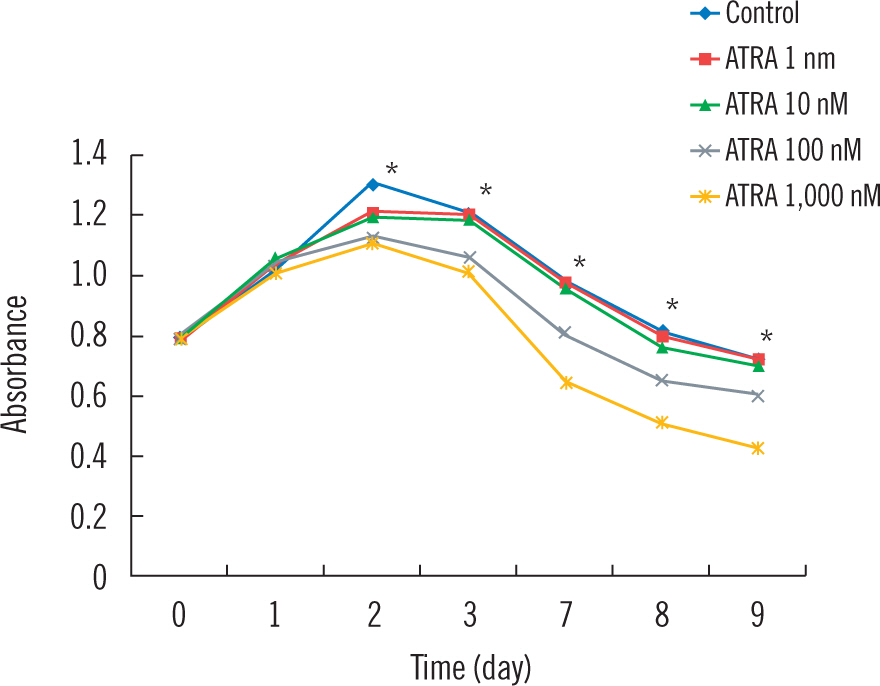
The proliferation rates of HUVECs in various concentrations of all-trans-retinoic acid (ATRA) from 1 to 1,000 nM. (*) Statistical difference with P<0.001 between every each group except between Control and ATRA 1 nM group, and ATRA 1 nM and ATRA 10 nM group.
In vitro proliferative HUVEC activity in ATRA with the wound healing promoter
The absorbances in the four groups (100 μg/mL PDRN, 1,000 μg/mL PDRN, 100 μg/mL PDRN with 1,000 nM ATRA, and 1,000 μg/mL PDRN with 1,000 nM ATRA) had increased on day 1 (Fig. 2 and Table 2). From day 4, the two ATRA added groups began to show decreased absorbance, and the decreasing tendency lasted until the end of experiment. Compared with the 100 μg/mL and 1,000 μg/mL PDRN groups, the ATRA added groups showed significantly decreased absorbance (P<0.01). There was also a statistically significant difference between the two ATRA groups (P<0.01). However, there was no statistically significant difference between the PDRN only groups.
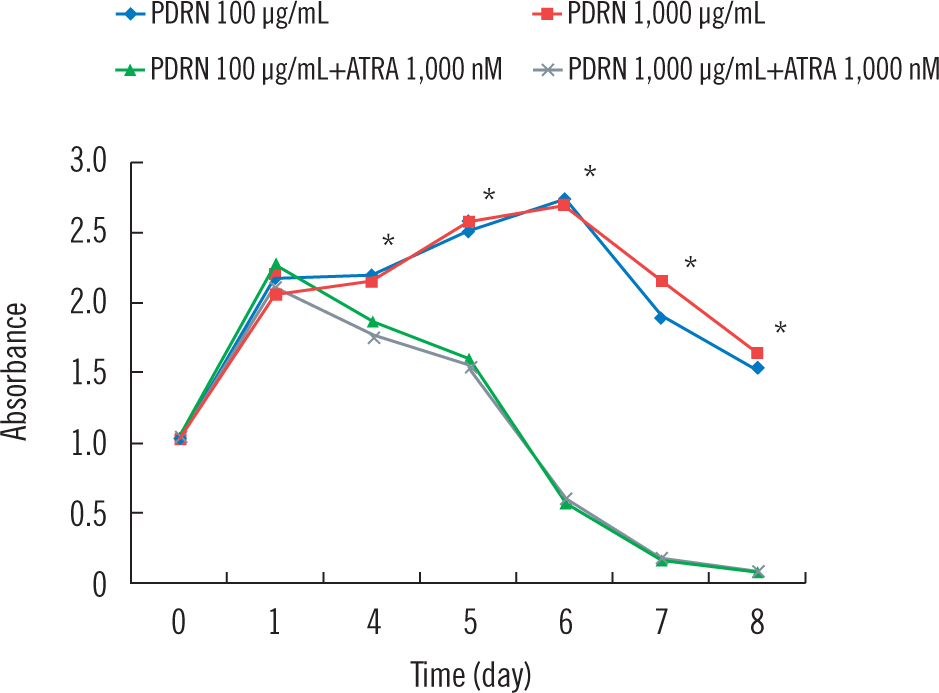
The proliferation rates of HUVECs in the presence of polydeoxyribonucleotide (PDRN) and 1,000 nM all-trans-retinoic acid (ATRA). (*) Statistical difference with P<0.01 between every each group except between PDRN only groups.
Discussion
Although the effects of retinoic acid in wound healing have been discussed over 50 years, its exact effectiveness has not yet been established. Current studies have reported that retinoic acid acts on two types of nuclear receptors: retinoic acid (RARs) and retinoid X (RXRs). By binding as ligands to RARs and RXRs, retinoic acid promotes transcription of the downstream target gene into mRNA and protein, which induces epithelial proliferation and differentiation [12,13]. There is also a report that retinoic acid affects growth factors such as transforming growth factor-β (TGF-β) and insulin-like growth factor-1 (IGF-1) that induce fibroblast activation and angiogenesis [2]. However, Karadag et al. [14] reported that isotretinoin treatment had decreased the level of IGF-1 and insulin-like growth factor binding protein-3. Hung et al. [9] demonstrated that continued retinoic acid had delayed the wound healing process by prolonging the inflammatory phase and inducing capillary dilatation, intercellular space widening, and a disorganized, noncohesive epithelium. There are also studies about tretinoin’s suppression of fibroblast activation; this activation had been suppressed by 10−3 mol/L and 10−5 mol/L of tretinoin, and concentration-dependent suppression of fibroblast activation was observed in the range from 10−11 to 10−6 mol/L tretinoin [15–17].
In this study, we analyzed the effects of retinoic acid on vascular endothelial cells, which are also critical in wound healing along with fibroblasts. The roles of vascular endothelial cells include producing nitric oxide and controlling platelet aggregation, fluid filtration, and particularly angiogenesis in wound healing. Currently, there are few studies about the effects of retinoic acid on endothelial cells. Uruno et al. reported the increase of nitric oxide production with ATRA in various types of endothelial cells, and Saito et al. reported that ATRA binding to RARs induced in vitro angiogenesis by increasing vascular endothelial growth factor (VEGF), hepatocyte growth factor, and angiopoietin-2 secretion [3,18]. However, Salazar et al. reported that treating endothelial cells with isotretinoin produced significant changes in the expression of genes implicated in cell adhesion and in lipid metabolism and led to an exacerbating effect on atherosclerosis risk factors [8].
The human umbilical cord is one of the most important sources of vascular endothelial cells that maintain nearly all of the features of native vascular endothelial cells including the expression of endothelial cell-specific markers such as von Willebrand factor and endothelium-specific adhesion molecule CD31, and expression of receptors for growth factors such as VEGF, TGF-β, and fibroblast growth factor [19]. Using these characteristics, we checked the proliferative HUVEC activity in various concentrations of ATRAs.
The control group, without the ATRA, showed higher proliferative activity than any experimental groups, and the activity decreased with increased concentrations of the ATRA in the experimental groups. This result is similar to the concentration-dependent suppression of fibroblast activation we mentioned above. The fact that proliferative activity decreased from day 2 until day 9 suggests that ATRA suppresses vascular endothelial cells in the hemostasis, inflammatory, and also proliferative phases of wound healing. Considering that lipid metabolites such as lipoxin, resolvin, protectin, and maresin are promoters of inflammatory phase resolution, we can infer that the decreased proliferative activity of vascular endothelial cells may affect the lipid metabolism and prolong the inflammatory phase [20]. The decreased proliferative activity of vascular endothelial cells may also affect wound vascularization in all phases of the wound healing process. Given that neoangiogenesis starts with angiogenic factors binding to endothelial cell receptors followed by endothelial cell proliferation and migration, decreased proliferative activity of vascular endothelial cells may interfere with neoangiogenesis and delay wound healing, which we confirmed in our next experiment with PDRN.
PDRN is a compound formed by polymers of deoxyribonucleotides with chain lengths ranging between 50 and 2,000 base pairs that affects cell proliferation mediated by the activation of purinergic receptors of the A2 subtype and the activation of salvage pathways [21]. PDRN derivatives could act as growth promoters for fibroblasts, osteoblasts, and endothelial cells and could stimulate nucleic acid synthesis [22,23]. As we added the PDRN in the HUVECs, the proliferation increased until day 6. However, the groups with PDRN and 1,000 nM ATRA showed decreased proliferative activity beginning on day 4 that lasted until the end of the experiment regardless of the PDRN concentrations. Therefore, we suggest ATRA as a powerful suppressor of HUVECs that furthers the wound healing process.
There are some limitations in this study. As there was no significant difference of HUVEC proliferation activity between the 1 nM ATRA group and 10 nM ATRA group, more delicate study design and repeated experiments are required to determine their exact suppressive and weak acting ranges. Recently, low-dose isotretinoin is recommended in certain settings, and association between isotretinoin and problematic skin healing after surgery is being challenged [24,25]. An additional study regarding the ATRA concentrations from 0 to 10 nM and adding lower doses of ATRA to HUVECs with PDRN would give a solution to this issue. Vascular endothelial cells are known to participate in wound healing by proliferation and migration. Because we focused on their proliferative characteristic in this study, our challenge is to focus on ATRA’s effects on endothelial cell migration in the next stage and ultimately, in vivo confirmation would lead to various clinical applications including in wound healing.
In conclusion, ATRA in concentrations from 10 nM to 1,000 nM has definitely suppressed the proliferation of vascular endothelial cells in vitro. Because vascular endothelial cells play a key role in coagulation, hemostasis, and angiogenesis in wound healing, retinoic acid may delay wound healing in such conditions.
Notes
No potential conflicts of interest relevant to this article are reported.

