 |
 |

|
| Aims and Scope |
| About the Journal |
| About the Society (KWMS) |
| Editorial Board |
| Open Access |
| Principles of Transparency and Best Practice |
| Editorial Office |
| Subscriptions |


|
| Aims and Scope |
| About the Journal |
| About the Society (KWMS) |
| Editorial Board |
| Open Access |
| Principles of Transparency and Best Practice |
| Editorial Office |
| Subscriptions |

AbstractFull-thickness burns almost always require skin grafting because the severity prevents the natural regenerative process. Here, we illustrate the potential to promote natural skin regeneration utilizing growth factors and regenerative proteins in purified amniotic fluid (PAF). A 24-year-old female suffered from full-thickness burns to her legs resulting from a rogue firework. This initiated a treatment plan involving extensive rehabilitation and skin grafting. Instead, the patient was treated with PAF (4 mL) administered topically over 2 days. Forty-five minutes following application, her pain decreased from 10/10 to 2/10. After two treatments her overall recovery time decreased by 75%–leaving her physicians surprised at the obviated need for skin grafting. Topical PAF relieved pain within minutes, making it easier to clean the affected area, enhancing sleep, and allowing more range of motion and autonomy. The efficiency of PAF to reduce pain allowed the patient to discontinue the use of prescribed hydrocodone, an opioid that is fueling an epidemic of abuse and addiction. Accelerated recovery time allowed the patient to return to work sooner, having broader economic implications on medical costs and personal time off. Finally, PAF enhanced the body’s own capacity to heal itself, avoiding painful and costly grafting and decreasing additional donor site scarring.
IntroductionSkin plays many important roles in regulating water and temperature balance, cell signaling, and hormone and immune peptide production and activation [1]. Therefore, basic elements–cells, growth factors, and matrix–should be used to enhance natural skin regeneration following injury [2]. Here we present the use of purified amniotic fluid (PAF) to improve burn wound healing time, obviate the need for skin grafting and reduce scarring. PAF contains over 300 bioactive proteins and extracellular vesicles (EVs) that contribute to the natural phases of skin regeneration [3].
Skin regeneration is a process involving three to four overlapping phases: hemostasis (coagulation), inflammation (mononuclear cell infiltration), proliferation (angiogenesis), and maturation (collagen deposition) [4-6]. The first most pivotal phase is the inflammation phase, which is regulated by macrophages. Macrophages have two distinct phenotypes, a pro-inflammatory M1 phenotype and an anti-inflammatory M2 phenotype. The role of EVs in cutaneous injury repair is to promote the critical switch in the phenotype of macrophages toward the anti-inflammatory, pro-regenerative M2 type [4]. EVs have the capacity to induce this switch through a plethora of intracellular messages in the form of anti-inflammatory and pro-regenerative factors, listed in Table 1.
The second pivotal phase in skin regeneration is the proliferation phase, where neo-angiogenesis (formation of new blood vessels) occurs. In this phase EVs become integral because they contain cargo rich in proteins, RNAs and microRNAs that induce angiogenesis (Table 1) [7]. Skin regeneration is a highly metabolic process and requires increased oxygen and nutrients, making it essential to restore the blood supply by promoting new blood vessel formation [7].
There are currently few standard-of-care options for severe burn patients. Full-thickness burns generally result in hypertrophic scarring and contracture, leading to disfigurement, and long-term disability caused by reduced range in motion [8]. The standard-of-care for less severe cases is typically as follows: the wounds are cleaned, debrided, dressed with analgesics and left to heal on their own–causing scars [9,10]. In more critical cases, the patient is referred for surgical evaluation to a burn specialist, who then performs biopsies or skin grafts–causing additional scars [11]. The lack of alternative treatment options opens the door for novel research to help promote the natural mechanism of wound healing. PAF and the EVs associated with it are natural sources of growth factors, specifically designed by nature to modulate inflammation, promote tissue repair and remodeling, and manage pain [12]. Herein we report the efficient and effective promotion of skin regeneration in response to topical application of PAF. This patient provided written informed consent for publication of this case report.
CaseA 24-year-old female was referred to our group after sustaining full-thickness burns on her right posterior thigh and superficial and deep partial-thickness burns on her lower legs and back. Her injuries resulted from an explosion while attending a 4th of July party where fireworks were being ignited. She was standing roughly 15 m away from where the fireworks were being ignited. As the fireworks were set off, the bundle came apart and the rockets flew at a horizontal angle into the crowd. While the patient began to run in the opposite direction, one of the rockets hit her and exploded. She fell to the ground while a bystander helped put out the fire that had ignited on her person. She was promptly rushed to the local emergency room.
The emergency room physician informed the patient that if not for the quick response, the severity of her injuries indicated possible loss of her right leg (Fig. 1A). The injuries to the right leg were cleaned and bandaged and she was sent home with an antibiotic cream and wrap and 5 mg of hydrocodone every 6 hours for pain management (Fig. 1B).
Upon waking the following morning, the patient was unable to move her leg due to severe pain. The patient reported a pain score of 6-8/10 during periods of immobility and higher when mobile. Because of the severity of the pain, the patient was taking more than the prescribed amount of hydrocodone (5 mg every 4 to 5 hours instead of every 6 hours). In addition, during wound cleanings the patient reported pain as “excruciating” with a score of (+10/10) and would require 10 mg hydrocodone prior to cleanings.
On the third day following the accident, the patient was referred to the burn unit where her legs were both debrided. Although the patient was a young and healthy individual, after assessing her injuries, the burn unit estimated her total recovery time (through secondary intention healing) to be greater than 2 months; skin grafting options were discussed for the full-thickness injuries located on the posterior right leg (Fig. C1, illustrated by arrows).
In an attempt to find an alternative to skin grafting, the patient consulted for a second opinion. To improve angiogenesis and speed up skin regeneration, topical application of 2 mL PAF was suggested. The use of PAF as an investigational drug product is currently under review by the United States Food and Drug Administration. On day 4 following the accident, PAF was sprayed over the wounds and the injuries were covered using dry, sterile dressings including rolled gauze with a tubular elastic retainer net to hold it in place. The patient reported no stinging, burning or pain during application of PAF to the wound sites.
Within 30 minutes, the white portions of her wounds began turning red, a visual indicator of enhanced blood flow (Fig. 2A and B) [13]. Prior to application of the PAF the patient reported experiencing 10/10 on the pain score. Forty-five minutes following application the patient reported subsiding pain, scored as 2/10. At this point she was able to lay down and apply pressure to the wounds without any pain. The patient’s mobility also improved significantly as she was able to place her heels almost completely to the ground when standing. Within hours the patient was able to discontinue the use of hydrocodone (opioids), instead taking 600 mg of ibuprofen every 6 to 8 hours. That same evening the patient was able to sleep through the night for the first time since the accident.
The following morning, the patient woke with pain scored 2/10. Showering and cleaning, which on the previous day resulted in pain scored +10/10, now resulted in a score of 3/10. Mobility was further restored as the patient was now able to lower the leg completely with minimal discomfort, an act that previously caused a score of 8-10/10 on the pain scale.
On day 5 following the accident the patient received a second 2 mL dose of topical PAF (24 hours after the initial dose) (Figs. 2C, 3A). Again, she reported no discomfort upon application and within minutes was able to lower her foot completely flat to the ground.
The day following the second dose the patient further decreased use of ibuprofen allotting for 12 hours between dosing. The patient was able to shower without any medication, walk without incident and move more freely.
At her next appointment with the burn unit, where she was originally scheduled to undergo a skin grafting procedure, the physicians concluded skin grafting was no longer required due to her extensive recovery. Prior to treatment with PAF the burn unit team proposed the necessity of skin grafting and a 2-month recovery time, however after PAF treatment no grafting was actually needed, and her recovery time was reduced to less than 2 weeks. The patient was back to work 11 days after her accident (Fig. 3B). As a result of this treatment, significant skin repair and remodeling was observed as promptly as the 3-week mark following injury (Fig. 4).
DiscussionIn this case report, topical application of PAF accelerated the recovery and pain management of a full-thickness burn injury. In summary, two doses (2 mL each) of PAF, administered as a spray to the affected area, reduced pain from 10/10 to 2/10, induced angiogenesis, obviated the need for skin grafting, improved skin remodeling, diminished scarring, and reduced recovery time by 75%, all which permitted the patient to return to her daily activities much sooner than expected.
These outcomes suggest that factors in PAF, mainly messages reducing inflammation, inducing angiogenesis and promoting cutaneous regeneration, provide restorative requirements during the injury proliferative phase. Components such as vascular endothelial growth factor (VEGF) and several microRNAs (listed in Table 1) are active biomolecules in PAF and are known to restore blood supply via new blood vessel formation. Characterization studies of PAF have also revealed the existence of anti-inflammatory mediators, such as the interleukin (IL)-10, IL-1ra, IL-13, and IL-27 [14].
Reducing inflammation can certainly reduce pain, yet pain relief to the level that was attained in this case suggests there may be other factors contributing to pain management which merits further investigation. A recent study investigating the effects of local injections of amniotic fluid in 96 patients with mild to moderate trigger finger showed an average pain score decrease from 5.19 to 1.19 on a 10-point scale [15]. Of the patients with clinically notable symptom relief, 89% had a decrease in average pain score [15]. In this case report the patient was able to stop using hydrocodone shortly after treatment, which poses the question of whether PAF would make for an efficacious alternative to opioid prescriptions. The opioid epidemic in the United States is challenging, as millions of people misuse prescribed opioids each year and many die from overdosing on synthetic opioids annually. Pain management studies using PAF may help reduce or even abolish the use of prescription opioids for wounds and surgical sites.
Due to the limited availability of noninvasive treatments for injuries of this magnitude, amniotic fluid therapy provides a complete and natural alternative for treating complex injuries. The multifaceted phases towards recovery for an injury such as a full-thickness burn make it challenging to rely upon one single method as a treatment option. However, the simple application of PAF provided the basic factors necessary to drive the natural phases of cutaneous regeneration and recovery. This report demonstrates the feasibility and practicality of using PAF as a novel standard treatment in modern wound care. As treatment cost is estimated to be roughly 500 US dollars per 1 mL, PAF is also much more financially attainable than the alternative skin graft. Further research is warranted on the capacity of PAF to induce neo-angiogenesis for faster healing, to help avoid the need for amputation, to negate the need for skin grafting, to reduce scarring and improve overall quality of life.
Conflict of InterestAuthors associated with Neobiosis, LLC are employed by manufacturer of donated purified amniotic fluid. The authors declare no other conflict of interest. Fig. 1.Posterior view of the right leg. (A) Upon arrival to the emergency room (ER), minutes after the accident. (B) After assessment in the ER and application of antibiotic cream. Both photos taken within hours of accident. (C) Arrows indicating full-thickness burns showing subcutaneous infiltration. For reference, the size of the largest wound shown by the top arrow (posterior thigh) is roughly 5 cm in height and 3 cm in width. 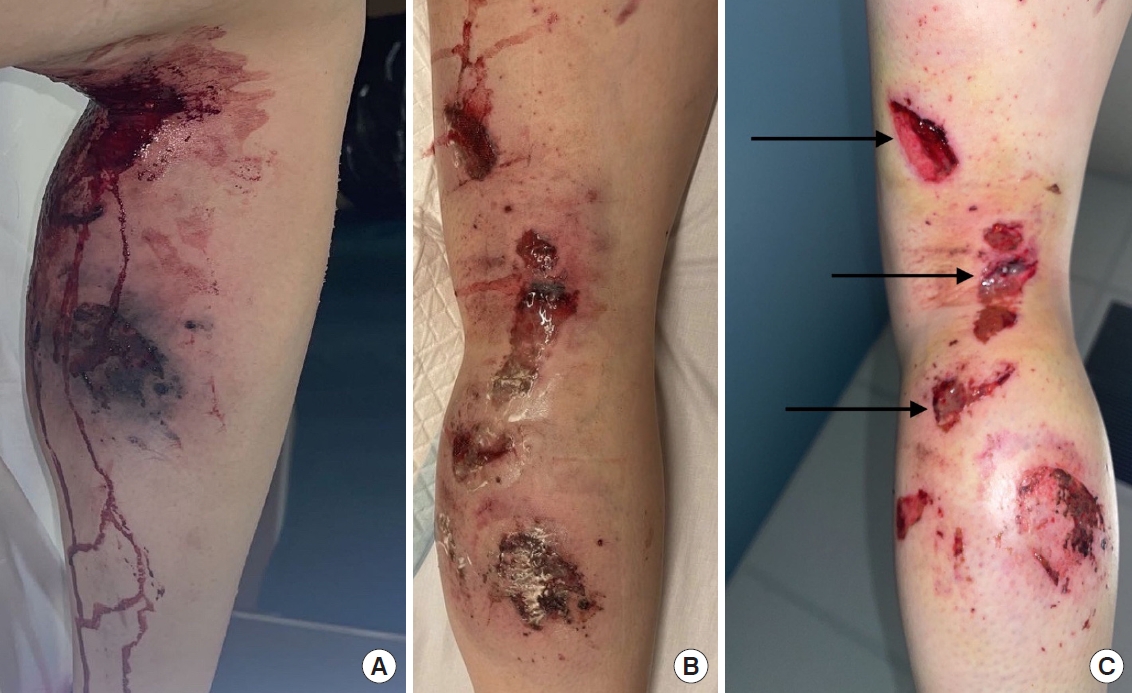
Fig. 2.Posterior view of right thigh. (A) Wound showing white interior, lacking vascularization, on the day of application of initial dose of purified amniotic fluid. (B) Wound 30 minutes after first application, showing red starting to fill in area, suggesting neo-angiogenesis. (C) Wound 24 hours after first application, already showing signs of cutaneous regeneration in the periphery of the injuries. 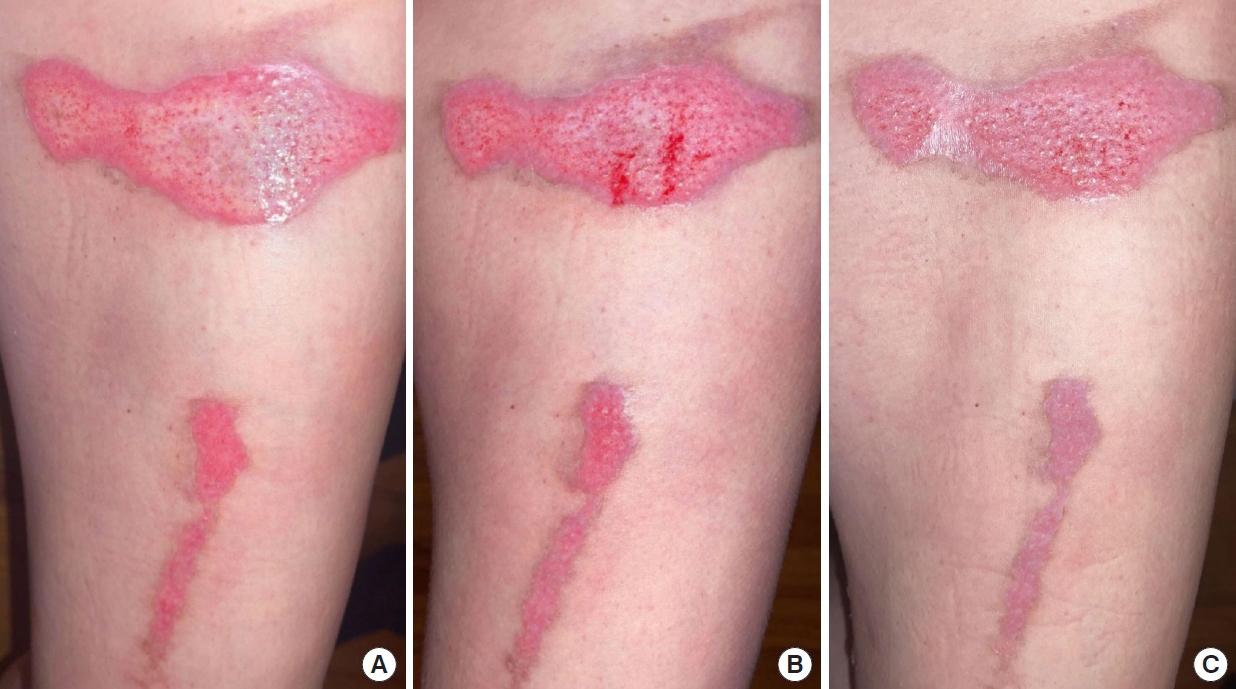
Fig. 3.Posterior view of the right leg. (A) Burn wounds on the day of application of initial dose of purified amniotic fluid, day 4 after the accident. (B) Wounds 1 week after purified amniotic fluid application. 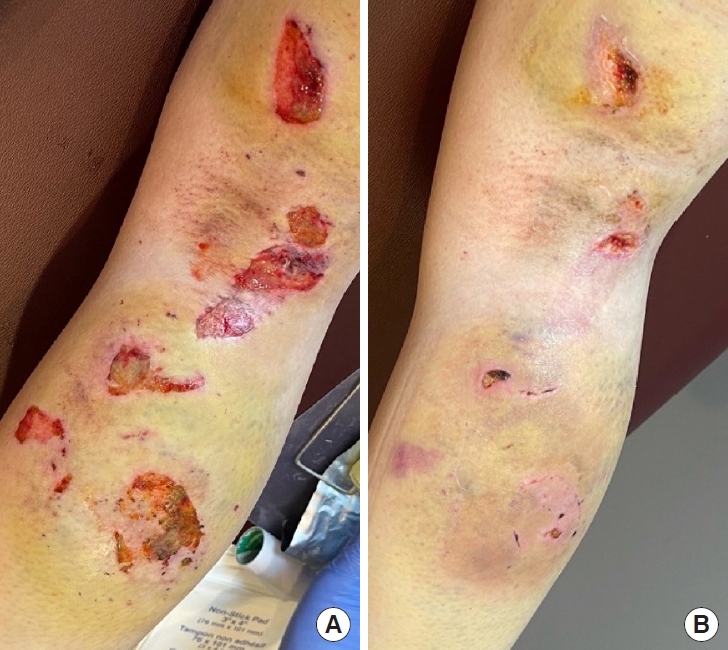
Fig. 4.Follow-up views of wounds. (A) Posterior right leg 3 weeks following injury. (B) Anterior thigh 3 weeks following injury. (C) Posterior right leg 6 months following injury. (D) Anterior thigh 6 months following injury. 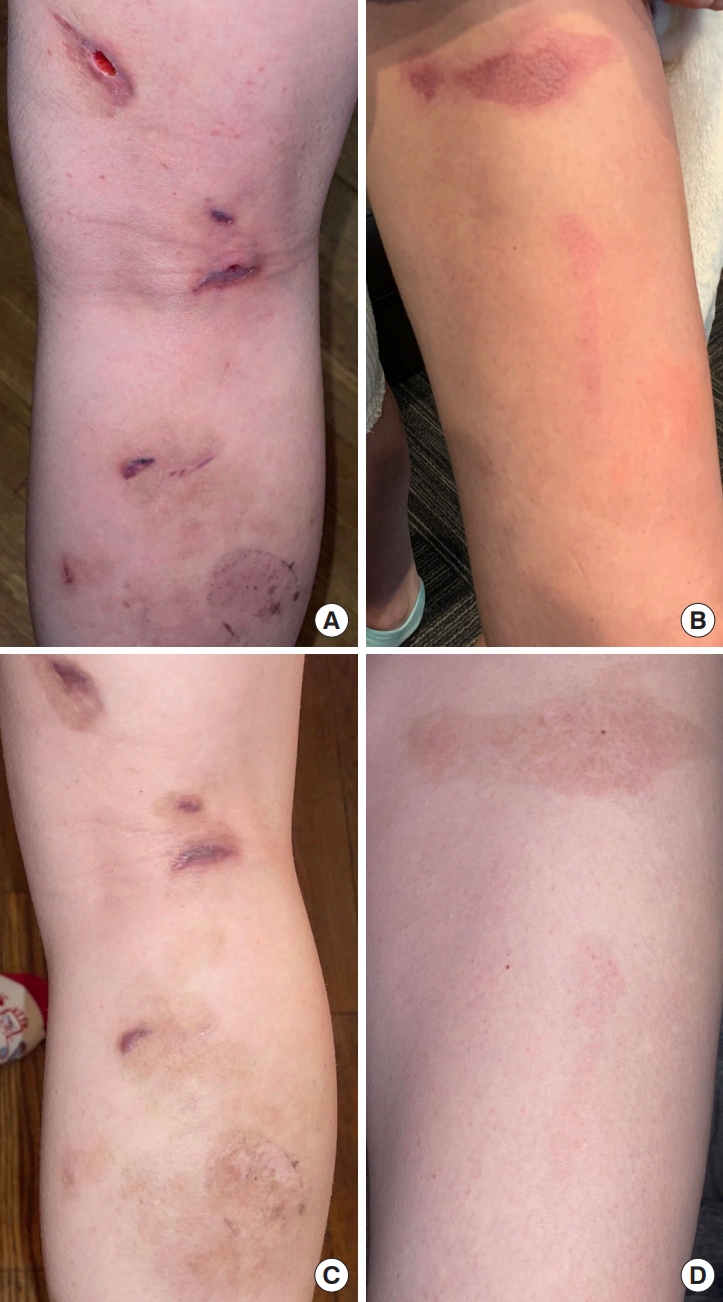
Table 1.Proteins and microRNAs detected in purified amniotic fluid that are known to help promote angiogenesis and cutaneous regeneration and remodeling References1. Nejati R, Kovacic D, Slominski A. Neuro-immune-endocrine functions of the skin: an overview. Expert Rev Dermatol 2013;8:581-3.
2. Shpichka A, Butnaru D, Bezrukov EA, et al. Skin tissue regeneration for burn injury. Stem Cell Res Ther 2019;10:94.
3. Hu P, Yang Q, Wang Q, et al. Mesenchymal stromal cells-exosomes: a promising cell-free therapeutic tool for wound healing and cutaneous regeneration. Burns Trauma 2019;7:38.
6. Sahoo S, Klychko E, Thorne T, et al. Exosomes from human CD34(+) stem cells mediate their proangiogenic paracrine activity. Circ Res 2011;109:724-8.
7. Landskroner-Eiger S, Moneke I, Sessa WC. miRNAs as modulators of angiogenesis. Cold Spring Harb Perspect Med 2013;3:a006643.
8. Brissett AE, Sherris DA. Scar contractures, hypertrophic scars, and keloids. Facial Plast Surg 2001;17:263-72.
9. Morgan ED, Bledsoe SC, Barker J. Ambulatory management of burns. Am Fam Physician 2000;62:2015-26.
10. Gacto-Sanchez P. Surgical treatment and management of the severely burn patient: review and update. Med Intensiva 2017;41:356-64.
11. Karlsson M, Elmasry M, Steinvall I, et al. Scarring at donor sites after split-thickness skin graft: a prospective, longitudinal, randomized trial. Adv Skin Wound Care 2018;31:183-8.
12. Morteza Bagi H, Ahmadi S, Tarighat F, et al. Interplay between exosomes and autophagy machinery in pain management: state of the art. Neurobiol Pain 2022;12:100095.
13. Kurkipuro J, Mierau I, Wirth T, et al. Four in one: combination therapy using live Lactococcus lactis expressing three therapeutic proteins for the treatment of chronic non-healing wounds. PLoS One 2022;17:e0264775.
|
|