Introduction
The fingertips are the human body area most frequently injured [1]. When reconstructing amputated fingertips, replantation by microvascular anastomosis is the treatment of choice. In situations where replantation cannot be performed, several other options are available, including stump revision, skin or composite grafting, and local and free flaps. If no suitable vessels for anastomosis are available, composite grafting could be a good option for restoring aesthetic appearance, sensation, and function of the fingertip without multiple time-consuming procedures. However, the success rates of composite grafting are highly variable and often unpredictable [2-4].
A moist environment is well known to be an important factor in wound healing [5]. Moist treatment of wounds enhances wound healing, boosts re-epithelialization, lowers rate of infection, promotes autolytic debridement and reduces scarring. On top of all these benefits, moisture prevents wound desiccation. In addition, cytokine and growth factors trigger beneficial effects and promote angiogenesis and revascularization [6-12].
A wet environment around a wound prevents desiccation, and thereby prevents necrotic tissue from developing on the surface of the wound. We therefore assumed that a dry environment would promote graft desiccation and increase the incidence of graft necrosis. Before revascularization, the graft is nourished by plasma diffused from the wound bed [13]. The longer the graft is protected from desiccation, the further will the host vessels from the wound bed grow into the graft, increasing the likelihood of survival for the graft. In the authors’ experience, conventional compression dressing with petroleum gauze does not guarantee adequate hydration, often resulting in desiccation of grafts and then graft necrosis. In this article, we introduce a new dressing method to maintain moisture, prevent desiccation of grafts and ultimately increase their chances of survival.
Methods
We conducted a retrospective chart review of patients who underwent composite graft of the fingertip between February 2012 and February 2019. The study was performed in accordance with the principles of the Declaration of Helsinki. As a retrospective review of medical records, the study itself did not require informed consent; written informed consent was obtained from the patients for the use and publication of their images. Patients who were injured proximal to the distal interphalangeal joint, those with bone exposure, contaminated wounds, autoimmune disease and those receiving dialysis were excluded. The following items were investigated: age, sex, site of injury, size of defect, mechanism of amputation, any documented complications, and the degree of graft survival.
Mechanisms of amputation were recorded as laceration, crushing or avulsion. Lacerations were defined as injuries from sharp objects, without tissue loss. Crushing was from blunt objects with some loss and crushing of tissues. Avulsion injuries were those with severe crushing or avulsion of tissues [14]. Graft survival was also grouped into three categories: complete, incomplete, and failed. Incomplete graft survival was defined as any graft on which there were patches of necrotic tissue interspersed with viable tissues. Failed graft was defined as total tissue necrosis at the tip of the finger [15].
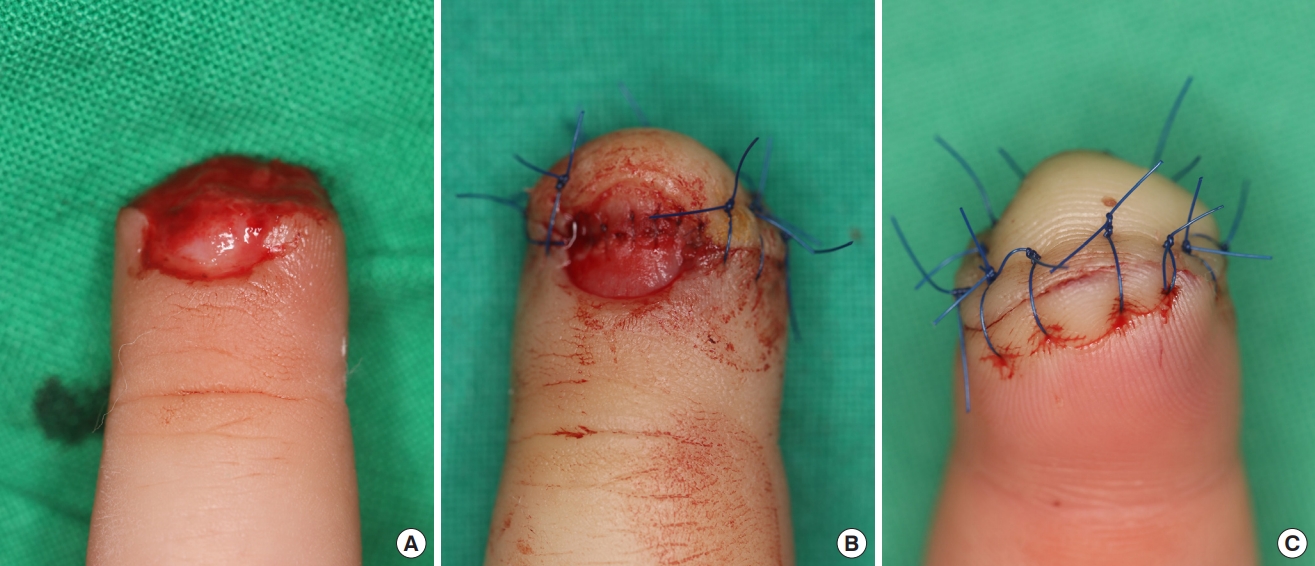
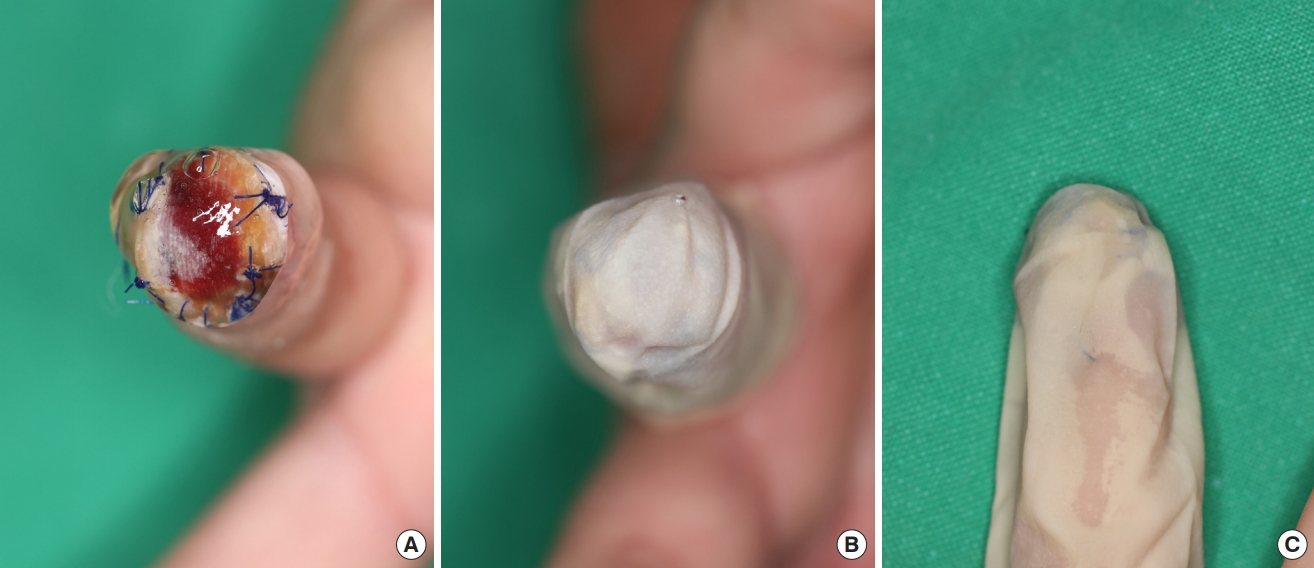
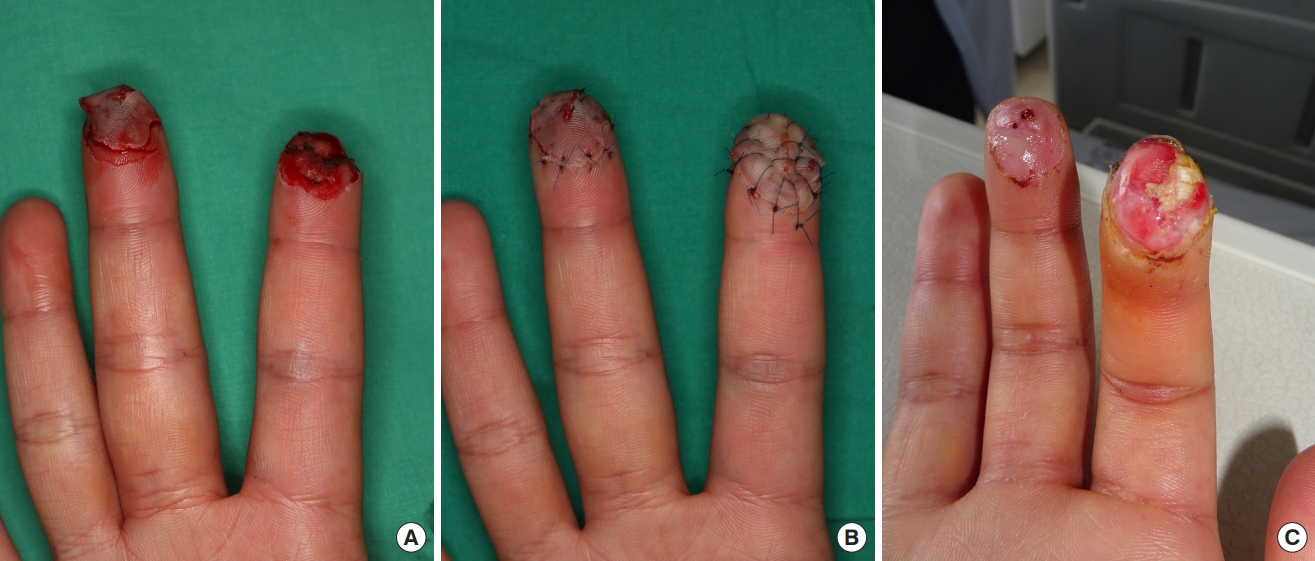
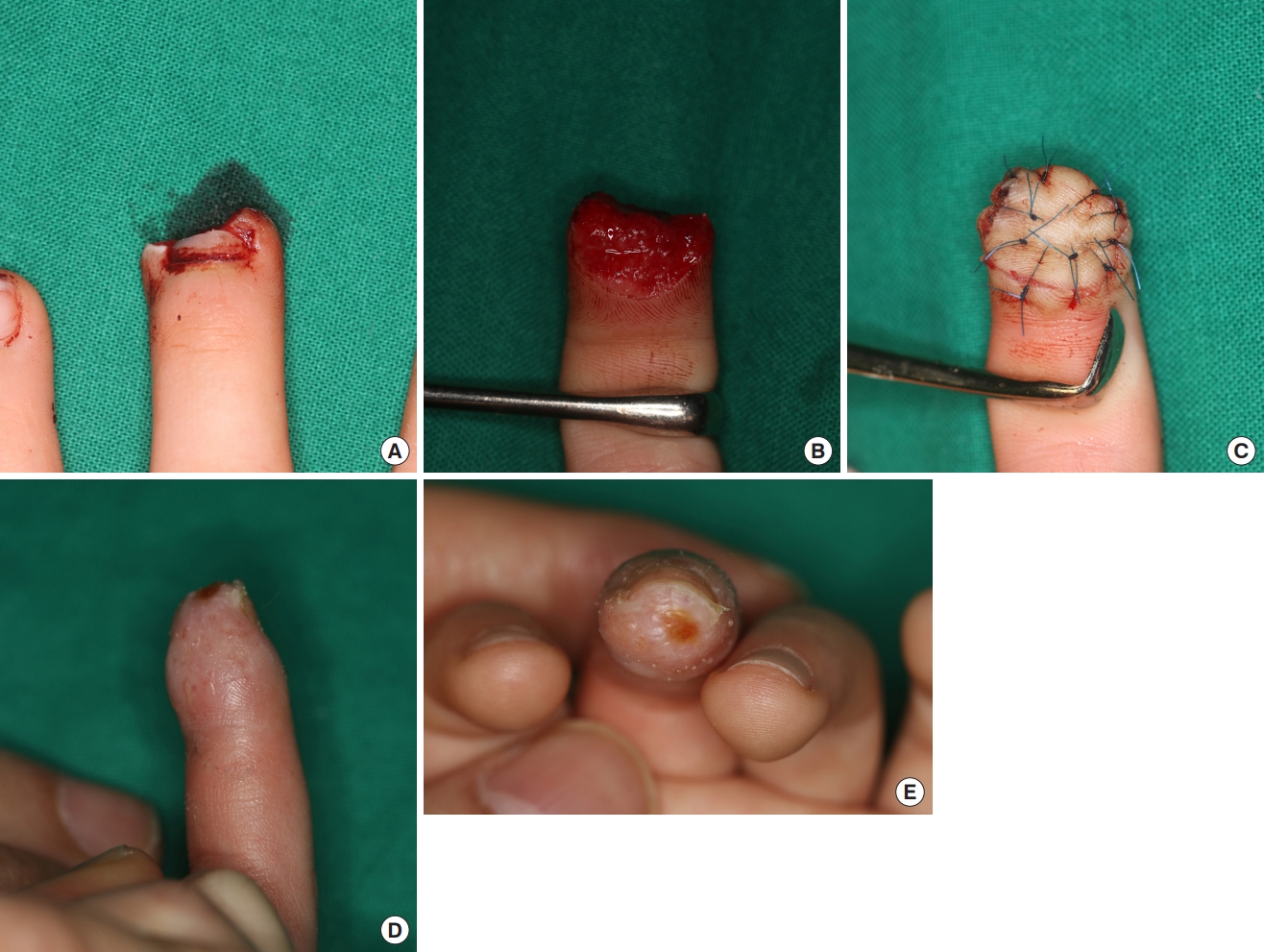
For all the patients, fingertip composite grafting was performed under digital block of the affected digit using 2% lidocaine. After applying a finger tourniquet, the wound and graft were irrigated thoroughly. Foreign bodies and devitalized tissues were debrided. The size of the defect was measured intraoperatively in length and width. After electrocauterizing to control bleeding, the amputated part was reattached as a composite graft using 5-0 nylon (Fig. 1). Duoderm Hydroactive Gel (Convatec, E.R. Squibb & Sons Inc., Princeton, NJ, USA) was then applied on the graft and covered with a “finger cap,” cut from a sterile latex glove (Fig. 2). After the composite graft was covered with the finger cap, the cap was held in place at the proximal end with gauze and elastic bandages to keep the gel from oozing out. The dressing, including the finger cap and hydrogel, was changed daily for the first 3 days after surgery. Afterwards, the frequency of dressing changes was adjusted depending on how well the graft was hydrated. The survival rates of the grafts were determined on the basis of the clinical photographs taken 4 weeks after the procedure (Figs. 3-5).
Results
Eighteen patients with 18 fingertips in total satisfied the inclusion criteria (Table 1). The patients’ mean age was 29±19 years (range, 2–58 years). Ten were male (56%) and eight (44%) were female. The most common mechanism of amputation was by crushing, observed in nine patients and the others were laceration in five patients and avulsion in four patients. The size of the fingertip defect ranged from 3×3 to 0.5×0.2 cm2, and the mean area of the defect was 2.18±1.99 cm2.
None of the cases showed any signs of inflammation or infection. In eight patients, full graft survival was observed. In all of the other 10 patients, the grafts survived incompletely, but the portion of the necrosis was insignificant (i.e., all necroses amounted to less than 50% of the size of the total defect). Healing by secondary intention was sufficient for all of them and no subsequent surgical procedures were necessary.
In 12 patients (67%), the defect was accompanied by a nail bed injury. None of them manifested complications or deformities related to the nail bed, including non-adherence, nail plate deformity, or hooked nail.
Discussion
In the 1960s, Winter first demonstrated the importance of a moist environment in wound healing [5]. Since then, the numerous benefits of wound hydration and moist wound healing have been proved and accepted as a basic principle in modern wound care. Moist wound treatment has various advantages: it boosts wound healing, enhances re-epithelialization, lowers rate of infection, promotes autolytic debridement, delivers good cosmesis by reducing scars, and allows prolonged presence of growth factors and cytokines which lead to favorable outcomes through wound contraction and angiogenesis promotion [5-12].
Revascularization is critical for graft survival. Before revascularization, the graft attains oxygen and nutrients through diffusion from plasma between the graft and the wound bed [16]. Revascularization starts as early as 2 days post-grafting and vessel growth occurs at about 5 μm/hr [13,17]. The thicker the grafts are, the harder it is for them to survive. Before angiogenesis is complete, graft survival purely depends on exudates from the wound bed. During this period, any desiccation will exacerbate graft necrosis and contribute to graft failure. Keeping the wound environment moist prevents a graft from desiccating and delays graft necrosis until the blood vessels are reconnected. The authors therefore hypothesized that a moist environment would eventually increase the graft survival rate. This had been demonstrated in the case of an upside-down split-thickness skin graft in a wet environment [18]. Even though the split-thickness skin graft was transplanted inverted (with the stratum corneum facing the wound bed), it could successfully survive if a wet wound environment was provided. In a dry wound environment, on the other hand, inverted grafts did not survive and showed significantly lower re-epithelialization. Zuhaili et al. [18] described that the successful outcome of the research owed to wound hydration and emphasized the importance of the wound environment in regeneration.
In our experience, when performing conventional dressings using topical antibacterial ointment and petroleum gauze, graft desiccation starts 5 to 10 days post-grafting. However, no graft desiccation was observed at 14 days after the procedures in all of the 18 cases. On the contrary, the grafts were hyperhydrated and became white and wrinkly. At first glance, hyper-hydration of the skin looks similar to moisture-associated skin damage (MASD). MASD is mainly due to chronic wound exudates containing protein-degrading enzymes, elevated proinflammatory cytokines and increased neutrophils [19,20]. Combined with proteases produced from bacteria, these exudates have corrosive effects on peri-wound skin [21]. The soft tissue defects observed in this study were grafted with the amputated parts in the acute stage and thus were different from the chronic wound environments characterized by prolonged inflammation which MASD is generally associated with. Besides, unlike the corrosive effect of MASD, hyper-hydration is limited to the stratum corneum. It is reversible and the skin itself is not excessively damaged [22]. In the authors’ experience, hyper-hydration due to continued exposure to the hydroactive gel over 2 weeks was harmless to the adjacent skin as well as the composite graft itself.
Although the authors are convinced of the effects of a moist environment in composite grafting, this study has some limitations. The absence of a control group made statistical comparisons impossible. In a single-center-based prospective study that included composite grafts in 57 digital tip amputations, 52.6% (n=30) of all the composite grafts survived completely. Partial survival and graft failure was 31.6% (n=18) and 15.8% (n=9) respectively. In that study, the authors concluded that smoking was the only significant factor that had a strong association with graft failure [23]. In a separate retrospective review of 100 patients under 16 years of age, 13% (n=13) survived completely, 46% (n=46) partially and 41% (n=41) failed [2]. Though a simple comparison of the present study and the former two studies would be far from significant, the fact that none (0%) of the grafts in the current study failed while 13% and 41% had failed respectively in the two previous studies might underscore the effectiveness of moist wound treatment on fingertip composite grafts, or it could be attributed to selection bias. In general, because it is difficult for thicker and wider composite grafts to survive, local, distant or free flaps are preferred over composite grafts for larger defects. This results in a tendency to perform composite grafts only on smaller defects, which could have affected the outcome of the study. The authors plan to include a more controlled statistical analysis of the effects of a moist environment on composite grafts in a future study.
In a wet or moist wound environment under occlusive dressings, evaporation, loss of energy and desiccation are reduced [11]. Presence of growth factors and proteinases in fluid exudates is maintained, also contributing to the healing [24,25]. In cases of split-thickness skin grafts, exudates containing these nutrients and growth factors are sufficient to nourish the graft during serum imbibition. On the other hand, when it comes to composite grafts, the tissue is thick and it is difficult for exudates to reach the most superficial area of the graft. If the wound environment is wet, occluded chamber fluid reaches equilibrium with the wound exudates and contains concentrations of electrolytes, glucose and protein very similar to those in serum [25]. In the same way, if a moist occlusive environment is provided for the composite grafts, evaporation and energy loss are reduced, growth factors and nutrients are preserved and contribute to graft survival.
Compared to other parts of the body, the fingertip is a much more difficult area to dress because of its frequent movements in daily life and its own curvature. Using a finger cap tailored from sterile latex gloves which are readily available makes dressings much easier. Moreover, because the latex is waterproof, dressing once a day or every 2 or 3 days is enough to maintain adequate hydration of the graft. Such a dressing constitutes the “ideal,” according to the British National Health System, in that it is impermeable to water and bacteria, minimizes the frequency of dressing changes, can absorb and contain exudate, provides thermal insulation, does not leave particulate contaminants in the wound, does not traumatize the wound bed when removed, and provides pain relief and comfort when used. By virtue of the amorphous hydrogel, this dressing does not come into contact with the wound bed and thus leaves nothing in the wound and does not traumatize the wound or cause discomfort. Although the authors had not specifically surveyed subjective or objective data on patient discomfort, very few patients complained about pain.
Various autologous and recombinant growth factors have been introduced as enhancing the wound healing process, including vascular endothelial growth factor, transforming growth factor, platelet-derived growth factor, polydeoxyribonucleotide, and platelet-rich plasma. With these growth factors, angiogenesis and damage from ischemia-reperfusion injuries of the grafts are improved and the graft survival rate could increase. These recombinant or autologous growth factors used in conjunction with wound hydration could promote graft survival even further.
In wound healing, wound hydration is the single most important external factor; therefore hydrating composite grafts could prevent necrosis from graft desiccation and increase the rates of survival for grafts. In treating fingertip defects using composite grafts, dressing with hydrogel and a finger cap is useful, not only keeping grafts hydrated but also reducing the frequency of dressings, lessening patient discomfort, and preventing infections. We recommend this dressing technique for successful fingertip composite grafting.